Matrix adhesion proteins: Matrix adhesion proteins, the final class of extracellular matrix constituents are responsible for linking the components of the matrix to one another and to the surfaces of cells. They interact with collagen and proteoglycans to specify matrix organization and are the major binding sites for integrins.
1. Fibronectin: Fibronectin is the principal adhesion protein of connective tissues. Fibronectin is a dimeric glycoprotein consisting of two polypeptide chains, each containing nearly 2500 amino acids. Fibronectin are proteins that connect cells with collagen fibers in the ECM, allowing cells to move through the ECM. Within the extracellular matrix, fibronectin is often cross-linked into fibrils. Fibronectin has binding sites for both collagen and GAGs so it cross-links these matrix components. Fibronectins bind collagen and cell surface integrins, causing a reorganization of the cell's cytoskeleton and facilitating cell movement. Fibronectin are secreted by cells in an unfolded, inactive form. Binding to integrins unfolds fibronectin molecules, allowing them to form dimers so that they can function properly. Fibronectins also help at the site of tissue injury by binding to platelets during blood clotting and facilitating cell movement to the affected areas during wound healing.
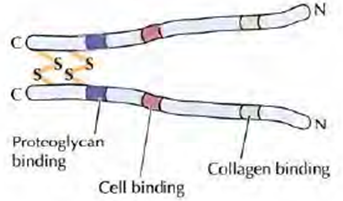
Figure 4: Structure of Fibronectin
2. Laminin: In almost all animals, Basal laminae contain distinct adhesion proteins of the laminin family. Laminins are heterotrimers of α, β and γ subunits which are the products of five α genes, four β genes, and three γ genes. Like type IV collagen, laminins can self-assemble into meshlike polymers. Such laminin networks are the major structural components of the basal laminae synthesized in very early embryos, which do not contain collagen. The laminins also have binding sites for cell surface receptors such as integrins, type IV collagen, and the heparan sulfate proteoglycan, perlecan. In addition, laminins are tightly associated with another adhesion protein, called entactin, which also binds to type IV collagen. As a result of these multiple interactions, laminin, entactin, type IV collagen, and perlecan form cross-linked networks in the basal lamina. They also support in cell adhesion.
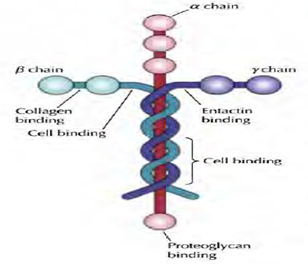
Figure 5: Structure of laminin