Collagen: In ECM of most animals, collagens are the abundantly found structural protein. In fact, collagen is the most abundant protein in the human body and accounts for 90% of bone matrix protein content. Collagens are present in the ECM as fibrillar proteins and give structural support to resident cells. Collagens are a large family of proteins containing at least 27 different members. They are characterized by the formation of triple helices in which three polypeptide chains are wound tightly around one another in a rope-like structure. The different collagen polypeptides can assemble into 42 different trimers. The triple helix domains of the collagens consist of repeats of the amino acid sequence Gly-X-Y. A glycine (the smallest amino acid, with a side chain consisting only of hydrogen) is required in every third position, so that the polypeptide chains can pack together close enough to form the collagen triple helix. Proline is frequently found in the X position and hydroxyproline in the γ position; because of their ring structure these amino acids stabilize the helical conformations of the polypeptide chains. The unusual amino acid hydroxyproline is formed within the endoplasmic reticulum by modification of proline residues that have already been incorporated into collagen polypeptide chains. Lysine residues in collagen are also frequently converted to hydroxylysines. The hydroxyl groups of these modified amino acids are thought to stabilize the collagen triple helix by forming hydrogen bonds between polypeptide chains. These amino acids are rarely found in other proteins although hydroxyproline is also common in some of the glycoproteins of plant cell walls.
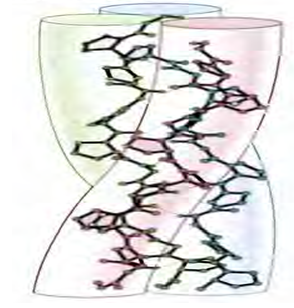
Figure 3: Structure of triple helix collagen
The collagen can be divided into several families according to the types of structure they form:
Table 1: Classification of collagens
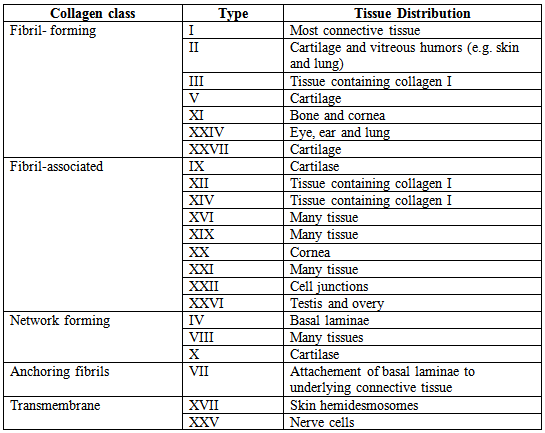
The most abundant type of collagen (type I collagen) is one of the fibril forming collagens that are the basic structural components of connective tissues. The polypeptide chains of these collagens consist of approximately a thousand amino acids or 330 Gly-X-Y repeats. After being secreted from the cell these collagens assemble into collagen fibrils in which the triple helical molecules are associated in regular staggered arrays. These fibrils do not form within the cell because the fibril forming collagens are synthesized as soluble precursors (procollagens) that contain nonhelical segments at both ends of the polypeptide chain. Procollagen is cleaved to collagen after its secretion, so the assembly of collagen into fibrils take place only outside the cell. The association of collagen molecules in fibrils is further strengthened by the formation of covalent crosslinks between the side chains of lysine and hydroxylysine residues. Frequently, the fibrils further associate with one another to form collagen fibers, which can be several micrometers in diameter.
Elastin: In contrast to collagens, Elastins give elasticity to tissues, allowing them to stretch when needed and then return to their original state. This is useful in blood vessels, the lungs, in skin, and the ligaments. Elastins are synthesized by fibroblasts and smooth muscle cells. Elastins are highly insoluble, and tropoelastins are secreted inside a chaperone molecule, which releases the precursor molecule upon contact with a fiber of mature elastin. Tropoelastins are then deaminated to become incorporated into the elastin strand. Diseases such as cutis laxa and Williams syndrome are associated with deficient or absence of elastin fibers in the ECM.