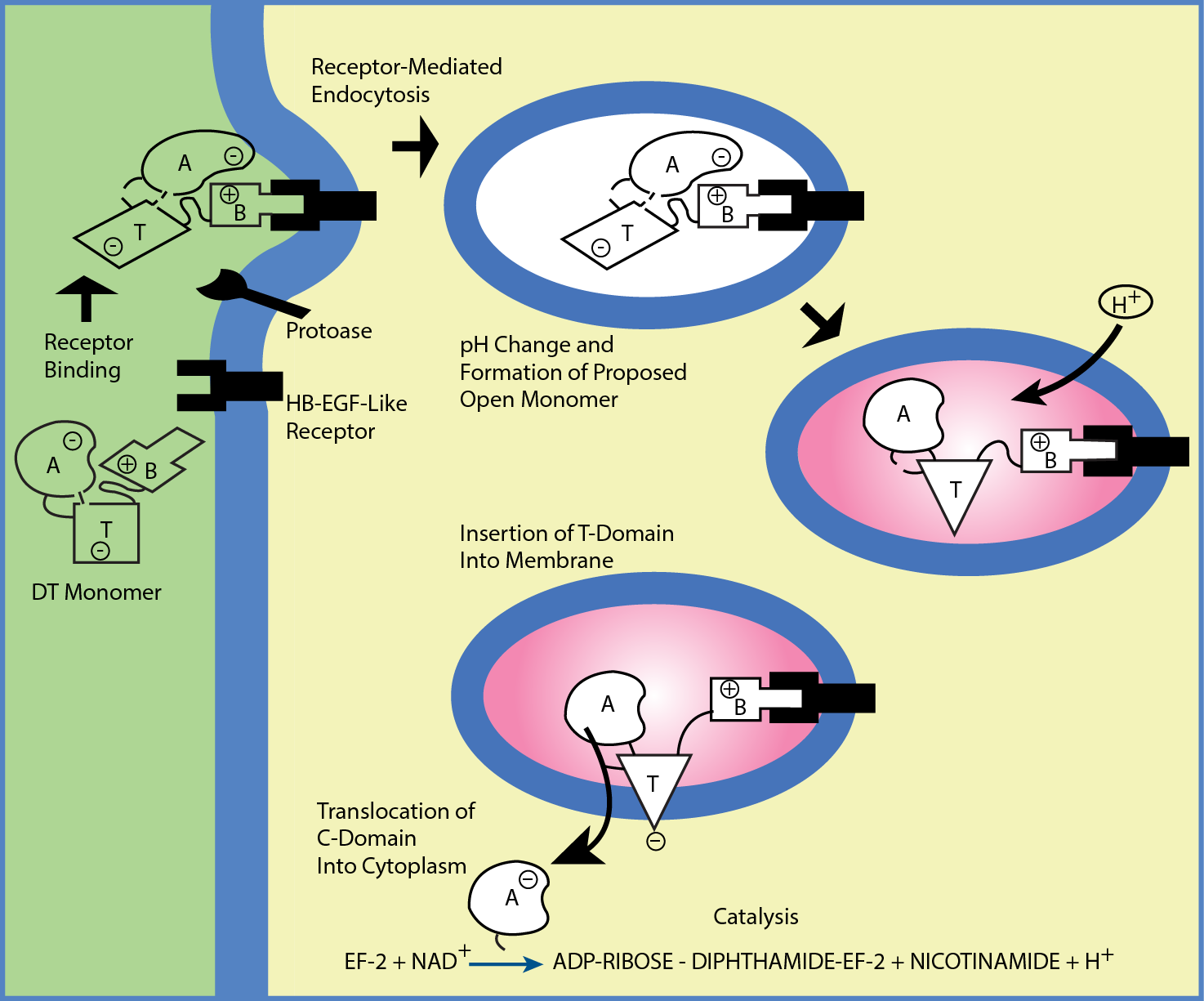
Figure 2: Entry and activity of diphtheria toxin (Dtx) in susceptible cells. The B domain of the toxin binds to a cognate receptor on a susceptible cell. The toxin is taken up in an endosome by receptor mediated encocytosis. Acidification of the endocytic vesicle allows unfolding of the A and B chains exposing the hydrophobic T domain of the toxin. The T domain inserts into the endosome membrane translocating the A fragment into the cytoplasm where it regains its enzymatic configuration. The enzymatic A component utilizes NAD as a substrate. It catalyzes the attachment of the ADP-ribose portion of NAD to elongation factor (EF-2) which inactivates its function in protein synthesis.
Clathrin independent endocytosis of ricin and Shoga toxin:
Plant toxin ricin binds to both glycolipids and glycoproteins with terminal galactose all over the cell surface and is therefore localized to all types of membrane invaginations and the toxin is internalized by all endocytic mechanisms. Ricin has been localized in clathrin-coated pits, but is still endocytosed when this pathway is blocked.
Clathrin-independent endocytosis is different from uptake by caveolae and macropinocytosis. For instance, clathrin-independent endocytosis occurs on the apical side of polarized cells, whereas caveolae are localized in the basolateral domain. Clathrin-independent endocytosis of ricin occurs when uptake from caveolae and clathrin-dependent endocytosis are inhibited by extraction of membrane cholesterol. Removal of cholesterol leads to the disappearance of caveolar and inhibits formation of invaginated clathrin-coated pits (Sandvig et al.,2000).