 |
Nuclear energy has been harnessed for power production, primarily through fission of Uranium, Thorium and Plutonium, with Uranium being the most common fissile material today. Looking at the binding energy curve, one can see that lighter nuclei have less binding energy than the heavier ones. Thus if a heavy nucleus breaks up into fragments, the sum of energies of the fragments would be less than the energy of the initial nucleus. This is the principle behind release of energy through fission. As the released energy is in MeV, fission released a million times more energy than chemical processes like burning of coal or oil which release energy of the order of electron volts. |
| |
Natural uranium consists of 98.275% of 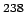 U, 0.72% of U, 0.72% of 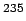 U and 0.005% of U and 0.005% of 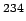 U. Most of the nuclear energy produced in the world today is produced by fission of U. Most of the nuclear energy produced in the world today is produced by fission of 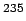 U. (An isotope of uranium U. (An isotope of uranium
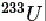 is also fissionable though it does not naturally occur but is produced by exposing thorium to neutrons.) The process of fission is initiated by capture of a slow neutron, called thermal neutron as the kinetic energy of such a neutron is of the order of thermal energy of air molecules, by uranium nucleus. Upon capuring a neutron, the uranium nucleus gets into an excited state and the delicate force balance inside the nucleus gets disturbed. The excited nucleus breaks up into two fragments of nearly comparable size. is also fissionable though it does not naturally occur but is produced by exposing thorium to neutrons.) The process of fission is initiated by capture of a slow neutron, called thermal neutron as the kinetic energy of such a neutron is of the order of thermal energy of air molecules, by uranium nucleus. Upon capuring a neutron, the uranium nucleus gets into an excited state and the delicate force balance inside the nucleus gets disturbed. The excited nucleus breaks up into two fragments of nearly comparable size. |