 |
Nuclear mass is usually expressed in terms of an unit known as the atomic mass unit , denoted by the letter u .One atomic mass unit is defined as one twelfth the mass of an atom of the most abundant isotope of Carbon , viz., 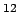 C C |
| |
In high energy physics, it is common to use mass and energy interchangeably and frequently one expresses mass in terms of MeV, which actually stands for the corresponding energy equivalent. In terms of the above units, the masses of a neutron and a proton are as follows : Mass of a neutron = 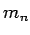 =1.008 665 u =1.008 665 u 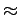
939.57 MeV/c 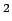 Mass of a proton = Mass of a proton = 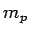 = 1.007 825 u = 1.007 825 u 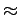 938.28 MeV/c 938.28 MeV/c 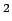 |