 |
The large binding energy suggests that the nucleons in an nucleus are very tightly bound. As a nucleus consists of protons which, being positively charged, would repel each other by Coulomb force, and neutrons which are charge neutral, the nucleons in a nucleus obviously cannot be glued together by electromagnetic forces which would actually make them fly apart. What binds the nucleons together is a new type of force, called the strong force . The characteristics of the strong force are |
|
the force is extremely short ranged - the force exists when the distance between nucleons becomes of the |
| |
order of nuclear dimensions ( 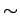 1 Fermi = 1 Fermi = 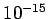 m). m). |
|
it is charge independent - the force is the same whether it is between a pair of protons or a pair of neutrons or |
| |
between a proton and a neutron. |
|
It is strong and attractive , relative to electrmagnetic forces it is about a hundred times stronger. |
| |
Inside a nucleus there is a competition between the repulsive Coulomb force and the attractive strong force. In most nuclei the strong force dominates. However, for heavier nuclei, there is a delicate balance between the two which can be easily disturbed leading to a fission of such nuclei. Spontaneous fission is of rare occurrence as the half life for fission is very high. The way to effect fission in a nucleus is to excite the nucleus - the energy required to initiate fission being between 5 to 6 MeV. |