38.8 Experiment 7: Complexometric Titration: Determination of hardness (Ca2+ ) of water using EDTA - complexometry |
Theory : |
| Hard water is one which does not easily lather with soap. This is due to the formation of precipitates of Ca and Mg stearates. |
|
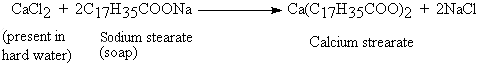
|
(38.8) |
|
|
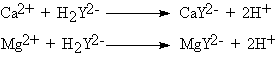
| Calcium and magnesium salts in water may be determined by a complexometric titration with ethylenediamine tetraacetic acid (EDTA, H4Y) which forms 1:1 complexes with most metal ions. Usually disodium salt of EDTA is used. The end points are obtained by using indicators appropriate to the specific metals. For determining calcium and magnesium, the indicator Euriochrome blackT is useful. A small amount of magnesium is essential for the indicator to function and is usually added to a buffer solution. The reactions involved are : |
| |
|
(38.9) |
| The indicator also forms complexes with Ca2+ and Mg2+, the Mg - indicator complex being wine-red. The free indicator is blue in colour. The Ca-EDTA complex is more stable than the Mg-EDTA complex. The Mg-indicator complex is stabler than the Ca-indicator complex, but less stable than the Ca-EDTA complex. As EDTA is added to the buffered (pH = 10) solution of Ca and Mg salts in the presence of Erichrome black-T, EDTA reacts with free Ca2+ ions first and then with free Mg2+ ions, if any, and finally with the Mg indicator complex. The end point is a sharp colour change form wine red to blue. The results are reported in mgs of CaCO3 per litre of the sample (ppm), irrespective of whether Mg was present or not. |
| |
s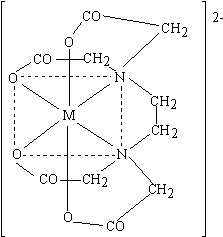
|
Figure 38.4 EDTA complex with a divalent metal |
| Procedure : |
| Prepare a 0.1% w/v (weight by volume) solution of calcium carbonate as a standard. Weigh out about 0.1 g of CaCO3 into the 100 mL volumetric flask. Add dilute HCl dropwise till effervevescence ceases and the solution is clear. Make up to the mark with distilled water. |
| Pipette out 10 mL of the solution into a conical flask and neutralize excess acid present by adding about 2 mL of ammonia solution and 5 mL of ammonia/ ammonium chloride buffer solution and 2-3 drops of indicator (Erichrome black-T). Titrate it against the EDTA solution (~ M/50), till the colour changes from wine-red to blue. |
| To determine the hardness of water, pipette out 50 mL of water sample into a conical flask, add 5 mL buffer and 2-3 drops of indicator and titrate it using EDTA solution. Report the hardness of water in ppm, i.e., parts of CaCO3 per million (mg/L) |
| In your report, explain how the choice of indicators for EDTA titrations is made in general, and list a few indicators used in EDTA titrations along with the metals they are used for and the pH at which the titrations are to be performed. |
| |
| |