When a base is added (volume, V) to acid, pH increases and reaches to 7.0 at the inflection point. Change in pH is very fast near the inflexion point. Therefore, value of
 pH / pH /
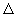 V is the highest at inflexion point. Hence, the end point of an acid base titration could be easily measured by a pH Meter. A representative curve for a pH-metric titration of a strong monobasic acid with strong base is shown below: V is the highest at inflexion point. Hence, the end point of an acid base titration could be easily measured by a pH Meter. A representative curve for a pH-metric titration of a strong monobasic acid with strong base is shown below: |