38.6 |
Redox Reactions |
| |
Oxidation-reduction reactions involve exchange of electrons between the reacting substances e.g., reaction between Fe2+ and Cr2O . . |
| |
6Fe2+ + 14H+ + Cr2O  6Fe3+ + 2Cr3+ + 7H2O 6Fe3+ + 2Cr3+ + 7H2O |
(38.4) |
| |
The above reaction is called as redox reaction because it could be represented by two half reactions involving oxidation and reduction, respectively. |
a) |
Oxidation of Fe2+ to Fe 3+ |
| |
Fe2+ Fe3+ + e- Fe3+ + e- |
b) |
Reduction of Cr 6+ to Cr3+ ; |
| |
Cr2O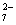 + 14H+ + 6 e- + 14H+ + 6 e- 2Cr3+ + 7H2O 2Cr3+ + 7H2O |
(38.5) |
| |
Equivalent weight of an oxidazing or reducting agent : |
| |
The equivalent weight of an oxidizing agent is the weight that accepts one mole of electrons and similarly the equivalent weight of a reducing agent is the weight that loses one mole of electrons. |
| |
This implies that in the reaction of ferrous ammonium sulphate with K2Cr2O7 in acidic medium, equivalent weight of ferrous ammonium sulphate is the same as its molecular weight. |
| |
Oxidation Reduction Indicator : |
| |
The change in potential at equivalence point of a titration may be detected by the addition of a suitable indicator. An indicator that exhibits a change in colour corresponding to a change in oxidation state is known as redox indicator. For example, colourless diphenyl-benzidine is oxidized to a coloured substance, diphenylbenzidine violet as shown: |
| |
 |
(38.6) |
| |
|