38.2 Experiment 1: Determination of the amount of sodium carbonate and sodium hydroxide in a mixture |
| Theory : |
Carbonate ion reacts with hydrogen ions in steps: |
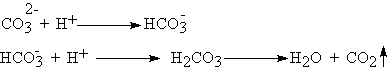 |
(38.1) |
|
|
The pKa1 and pKa2 values of H2CO3 are quite distinct and so when a carbonate solution is titrated against hydrochloric acid, there occur two distinct regions of sharp pH change. The first corresponding to the formation of HCO (pH 8 to 10) and the second due to complete neutralization at pH 4-6. The first is roughly in the pH range in which colour of phenolphthalein changes from red to colourless and the second is that at which methyl orange changes from yellow to orange red. This end point, however, is not very sharp in the titration of the strong base NaOH. The sharp change of pH occurs over a range of pH that includes the regions of colour change of both the indicators, so both of them give the end point correctly. (pH 8 to 10) and the second due to complete neutralization at pH 4-6. The first is roughly in the pH range in which colour of phenolphthalein changes from red to colourless and the second is that at which methyl orange changes from yellow to orange red. This end point, however, is not very sharp in the titration of the strong base NaOH. The sharp change of pH occurs over a range of pH that includes the regions of colour change of both the indicators, so both of them give the end point correctly. |
| |
| When we have both sodium carbonate and sodium hydroxide present together in a solution, a titration using phenolphthalein gives the titre (volume at the equivalence point) corresponding to sodium hydroxide plus half the carbonate and the titre obtained with methyl orange corresponds to the total alkali. The individual sodium carbonate and hydroxide concentrations may be calculated from the data. |
| |
| The HCl solution used may be standardized by titration with a standard solution of sodium tetraborate decahydrate (borax) or anhydrous sodium carbonate. The reaction involved in the case of borax is |
| |

|
(38.2) |
Borax is preferable as a primary standard because of its higher equivalent weight. |
| Procedure : |
| Prepare 100 mL of a standard solution of Na2B4O7 .10H2 O (approximately N/20) by weighing accurately about one gram of borax, dissolving it in distilled water and making up to 100 mL. Titrate 10 mL portion of this solution against the supplied hydrochloric acid till concordant titres are obtained, using 2 drops of methyl orange as indicator. Calculate the strength of the HCl solution. |
| |
| Pipette out 10 mL solution of a mixture of sodium carbonate and sodium hydroxide into a conical flask, add two drops of methyl orange indicator and titrate against HCl, till the colour changes from pale yellow to orange. Note the titre value (V1 ). Titrate 10 mL portions of the solution using phenolphthalein as indicator (1-2 drops). The color changes here at the end point is from red to colourless and is quite sharp. Let the titre be V2 of HCl. Therefore, 2(V1 - V2 ) corresponds to carbonate, and V1 2(V1 V 2 ) = 2V2 V1 corresponds to sodium hydroxide. Calculate the amount of NaOH and Na2CO3 present in a litre of the given solution in g/L. |
| |