To calculate the collision frequency, consider the motion of the molecule labeled A. In time
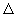 t, the molecule moves a distance t, the molecule moves a distance
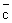 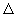 t where t where 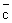 is the average speed of the molecule. Any other molecule which is within the volume
of the cylinder is the average speed of the molecule. Any other molecule which is within the volume
of the cylinder 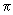  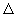 t d 2 spanned by molecule A, will collide with molecule A. Here t d 2 spanned by molecule A, will collide with molecule A. Here 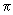 d 2 is the area of the cylinder spanned by A. This collision cylinder is shown in Fig 32.1. All the molecules that collide with A can be easily identified in the figure. During collisions, the directions of molecules change and the cylinder is not a rigid space but the effectively available space for the molecules. Since all molecules are moving, we need to use the relative speeds between molecules. From kinetic theory of gases, the average relative speed between molecules A and B is d 2 is the area of the cylinder spanned by A. This collision cylinder is shown in Fig 32.1. All the molecules that collide with A can be easily identified in the figure. During collisions, the directions of molecules change and the cylinder is not a rigid space but the effectively available space for the molecules. Since all molecules are moving, we need to use the relative speeds between molecules. From kinetic theory of gases, the average relative speed between molecules A and B is
|
| |
| 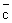 rel = (8 kBT / rel = (8 kBT / 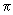 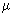 )1/ 2, )1/ 2, 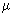 -1 = m A-1 + m B-1 -1 = m A-1 + m B-1
|
(32.2) |
Where 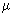 is the reduced mass between A and B. For identical molecules, is the reduced mass between A and B. For identical molecules, 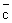 rel = rel =
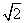 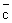 , where the latter is calculated with m in the denominator of Eq. (32.2) in place of the reduced mass. The factor 21/2 is because , where the latter is calculated with m in the denominator of Eq. (32.2) in place of the reduced mass. The factor 21/2 is because 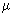 = m / 2. The area of the cylinder is called the collision cross section. Molecules outside this area do not collide with A. If NA is the number of molecules of A in volume V, the collision frequency is given by = m / 2. The area of the cylinder is called the collision cross section. Molecules outside this area do not collide with A. If NA is the number of molecules of A in volume V, the collision frequency is given by |
| |
z = 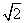 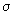 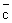 NA / V NA / V |
(32.3) |
| This z is the number of collisions encounter by a single molecule. Since there are NA molecules in volume V, the number of collisions between molecules of A in unit time and unit volume, ZAA |
| |
|
| ZAA = (1/2) z NA / V |
(32.4) |
| Substituting the values of all the factors, we have |
ZAA =
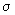 (4 kBT / (4 kBT / 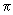 m )1/ 2 ( NA / V ) 2 m )1/ 2 ( NA / V ) 2 |
| If nA is the number of moles of A in volume V, then, [A] = molar concentration of A = nA / V . Since NA = nAL where L = Avogadro number, we have for Z AA |
| |
ZAA = 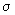 (4 kBT / (4 kBT / 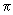 m )1/2 L2 [A] 2 m )1/2 L2 [A] 2 |
(32.5) |
| |
| |