The rate of the reaction, r = dP/dt, depends on the following factors: a) The concentrations or the number densities of molecules A and B, i.e., NA and NB ( NA = numbers of molecules in unit volume of the container, similarly NB ), b) number of collisions between of A and B in unit time. This is the collision rate. Larger the relative velocity between A an B, larger the collision rate.The relative speed between A and B is obtained from 1/ 2
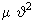 = 1/2 kBT or = 1/2 kBT or
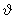 , the relative velocity = ( kBT / , the relative velocity = ( kBT /
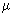 )1/2 is determined by the temperature T at equilibrium; and thirdly, c) the collision cross section, )1/2 is determined by the temperature T at equilibrium; and thirdly, c) the collision cross section,
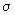 . The collision cross section is the area within which the center of A should be around the target B so that a collision can be said to have occurred. Let us first calculate the collision frequency between A and B. . The collision cross section is the area within which the center of A should be around the target B so that a collision can be said to have occurred. Let us first calculate the collision frequency between A and B.
|