Problems |
| |
| 32.1)
|
If z is the collision frequency, 1 / z may be thought of as the mean time before successive collisions. If we multiply the mean time with the mean speed, we get a length, which is now the mean or average distance a particle travels before undergoing a collision. This is called the mean free path Show that the mean free path for A - A collisions, 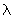 = = 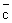 / z = (kBT / p ) / ( 2 1/ 2 / z = (kBT / p ) / ( 2 1/ 2 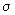 ) ) |
| |
| 32.2) |
Calculate the volume of the collision cylinder for sodium atoms at 300 K. Calculate the average velocity from
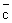 = (8 kT / = (8 kT /
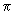 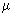 )1/2, where )1/2, where 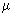 = m / 2 and m is the mass of a sodium atom. Use = m / 2 and m is the mass of a sodium atom. Use
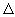 t = 100 ps (pico seconds) t = 100 ps (pico seconds) |
| |
| 32.3) |
The value of P for the reaction between NO and Cl2 giving NOCl and Cl is 0.018 at 298 K. If the experimental value of A is 4.0 * 109 L mol -1 s-1, estimate the collision diameter for the reaction. If the collision cross section of NO is 0.42 nm2, what is the collision cross section of Cl2? |
| |
| 32.4) |
The P factor for the harpoon mechanism between Na and Cl2 is 2.3. If the value of d for the reactants is
3.5
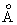 , what is the value of I - Ea for the reaction? Compute the value of I if Ea is 225 kJ / mol. , what is the value of I - Ea for the reaction? Compute the value of I if Ea is 225 kJ / mol. |
| |
| 32.5) |
Calculate the collision frequencies and collision densities at 298 K and 1 bar pressure for: a) pure N2, b)pure O2 c) equimolal mixture of N2 and O2. The collision diameters for N2 and O2 are 280pm and 300 pm respectively. 1 pm = 10 -12 m = 100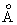 |
| 32.6) |
At 300 K, if the activation energy of the reaction is 15 kJ/mol, what fraction of collisions occur with the required kinetic energy? How does this fraction change at 1000K and 2000K? |
| |
| 32.7) |
Even though C2H4 and C6H6 are not spherical, collision diameters are used in calculations involving these molecules How can this be justified? |
| |
| 32.8) |
The collision cross sections in nm2 for atoms/ molecules are given in parenthesis below against the atoms / molecules He (0.21), Ar (0.36), O2(0.4), Cl2 (0.93), CO2 (0.52), CH4 (0.46), C2H6 (0.64), C6H6 (0.88). For some of these, try to estimate these values from the information on molecular / atomic sizes and bond lengths. |
| |
| 32.9) |
How can you calculate the mean free path (the average distance a molecule traverses before a collision) from mean velocity and the collision frequency? |
| |
| 32.10) |
Calculate the collision theory rate constant at 500K for the bimolecular reaction H2(g) + I2 (g)
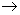 2HI(g). The activation energy is 170 kJ / mol. Use the reduced mass in your calculation. 2HI(g). The activation energy is 170 kJ / mol. Use the reduced mass in your calculation. |
| |
| 32.11) |
In the present lecture, we have verified the value of P for the H2 + C2H4 reaction. Verify the values of P for the reactions in the Table 32.1 involving reactions of NOCl and ClO |
| |
| |