| |
| Recap |
| In
this Lecture you have learnt the following |
| |
Summary |
| |
In the present lecture, we have been introduced to the collision theory of reaction rates. Collision frequency between reactants is calculated using the results of kinetic theory of gases. In a preliminary definition of a collision, two molecules are said to collide if the second molecule approaches the first molecule within an area called the collision cross section which equals
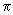 d2 where d is the average diameter of the two molecules. The rate constant is expressed as a product of the cross section, relative velocity and a Boltzmann factor accounting for the fraction of activated molecules. d2 where d is the average diameter of the two molecules. The rate constant is expressed as a product of the cross section, relative velocity and a Boltzmann factor accounting for the fraction of activated molecules. |
| |
|
| |
This simple collision theory departs significantly from experimental results. This is rationalized by noting that the reaction cross sections are different from collision cross sections. Most collisions (as exemplified using the collision between C2H4 and H2) do not occur with the correct orientations of the reactants. The effect of these orientations can be incorporated using a steric factor. The reaction cross section depends on relative velocities as well. This can be taken into account by including the experimental energy (or speed) dependent cross sections along with the Maxwell Boltzmann velocity distribution. In reactions where ionic interactions can contribute, the reaction cross section can be larger than the collision cross section. This was explained using the harpoon mechanism in the K + Br2 reaction. The experimental determination of reactive cross sections and their theoretical estimation is yet a challenge in chemical kinetics, but very significant progress has been made in the case of several elementary reactions. |
| |
|
| |
|
| |
Congratulations, you have finished Lecture 32. To view the next lecture select it from the left hand side menu of the page |
|
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
|