At large distances, neutral K and Br2 have lower energy than ionic K + and Br2- because more energy is needed to separate the ionic species. For all r greater than r*, the potential energy curve for the neutral reactants lies lower than the potential energy for the ionic reactants. At r = r*, the two potential energies are equal and for r values less than r*, the ionic potential energy curve is lower than the neutral reactants curve, because of the strong electrostatic attraction between K+ and Br2-. The difference in the energy (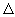 E) between the charged pair ( K + and Br2-) and the neutral pair is E) between the charged pair ( K + and Br2-) and the neutral pair is |
| |
| 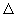 E = ( I - Ea - e2 / r ) - E neutral pair E = ( I - Ea - e2 / r ) - E neutral pair
|
(32.24) |
| |
Where I is the ionization energy of K, Ea, the electron affinity of Br2 and - e2/r, the electrostatic attraction. For
r < r *, 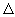 E is negative (more attractive) and therefore the distance at which the electron jumps from K to Br2 is when r = r *, where the term in brackets goes to zero, i.e., E is negative (more attractive) and therefore the distance at which the electron jumps from K to Br2 is when r = r *, where the term in brackets goes to zero, i.e., |
| |
| e2 / r * = I - E a or r * = e2 / ( I - Ea) |
(32.25) |
| |
Since the electron or the harpoon shoots across from K to Br2 when r = r*, 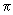 ( r*) 2 may be identified as the reaction cross section. It is after this transfer that the reactants are further accelerated towards each other and finally depart as products KBr and Br. This reaction cross section may be compared with the collision cross section ( r*) 2 may be identified as the reaction cross section. It is after this transfer that the reactants are further accelerated towards each other and finally depart as products KBr and Br. This reaction cross section may be compared with the collision cross section 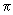 d 2 where d is the contact distance between K and Br2. This ratio is is the steric factor P discussed earlier. d 2 where d is the contact distance between K and Br2. This ratio is is the steric factor P discussed earlier. |
| |
P = 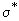 / / 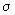 = ( r* / d ) 2 = { e2 / d ( I - Ea ) ]} 2 = ( r* / d ) 2 = { e2 / d ( I - Ea ) ]} 2 |
(32.26) |
| |
For K, I = 420 kJ / mol, Ea of Br2 is 250 kJ/ mol and d = contact distance = [ d (K) + d (Br2)] / 2 = 400 pm. d(K) is the diameter of K and d(Br2) is the average diameter of Br2. We note that this calculated value of P
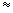 5 which matches with the experimental value of P. 5 which matches with the experimental value of P. |
| |
| To find the reaction cross section of reactions, one needs to know the potential energy curves (surfaces when more than one independent variable like r above are involved) and other details such as probabilities of passage from reactants to products. These involve detailed microscopic calculations. Experiments involve crossed molecular beams which will be outlined in a later lecture. |
| |
| |