We notice from the figure that at pressures between 102 and 105 Pa, explosions occur over 700 K. At a given temperature, say 800, K there is a pressure dependence too. Along the line ab at 600 K, explosion occurs only above P = 106 Pa. At 800K, along the line cdef explosion first starts around 103 Pa. Between 104 Pa to 5 * 105 Pa, there is a no explosion region and above 5*105 Pa, we have an explosion region once again. These variations are due to the dominance of different reactions in different temperature and pressure regimes. At the pressure at d (i.e., when the first explosion limit is reached), diffusion of the radicals to the end of the container is prohibited and the surface termination of radicals through reactions such as |
| |
| H  or O or O  or OH or OH  + surface + surface
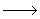 inert species (surface termination reaction) inert species (surface termination reaction)
|
(30.21a) |
| |
HO 2 + HO 2 + HO 2 + surface 2 + surface
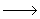 H2O + O2 + O (surface termination reaction) H2O + O2 + O (surface termination reaction) |
(30.21b) |
| is reduced and branching reactions dominate in the bulk of the container. As the pressure reaches e, in the region ef, gas phase terminations get an opportunity to reduce the radical populations through reactions with neutral gas species M present in the reaction mixture. |
M + H  + O2 + O2 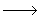 M + HO2 M + HO2 |
(30.22) |
| The HO2 radical is relatively quite stable. |
| Above the limit f, branching reactions and autocatalylic steps dominate (as the diffusion is considerably reduced) and one enters the explosive region once again. |
| |
| In several other explosive processes, similar underlying schemes are involved. Obtaining the complete rate expressions for these schemes is a challenging task as the approximations such as steady state or preequilibrium hardly apply and the number of reactions involved is also quite large. |
| |
| |