30.8 |
Problems |
30.1) |
Derive a rate law for the reaction scheme in equation (30.1) using the following steps. Obtain d [ CH 3 ] / dt from 3 ] / dt from |
| |
reactions involving k1, k2, k3 and k4 and d [  CH2OCH3] / dt in terms of k2 and k3. Set both to zero in the steady state approximation. Adding the two, get [ CH2OCH3] / dt in terms of k2 and k3. Set both to zero in the steady state approximation. Adding the two, get [  CH3] = { k1 [ CH3 OCH3 ] / 2 k4 } 1 / 2 . The rate of the reaction can be expressed in three ways as d [CH4] /dt or as d [HCHO] /dt or as CH3] = { k1 [ CH3 OCH3 ] / 2 k4 } 1 / 2 . The rate of the reaction can be expressed in three ways as d [CH4] /dt or as d [HCHO] /dt or as |
| |
- d [ CH3 OCH3 ] /dt. Show that all three lead to k2 (k1 / 2 k4)1 / 2 [ CH3OCH3] 3 / 2. Assume that the rate of initiation is much greater than the rate of propagation. |
| |
|
30.2) |
Obtain the steady state concentration of H and Br and Br in eq (30.8). in eq (30.8). |
| |
|
30.3) |
A branched chain reaction sequence may be written as A 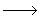 B; A + R B; A + R 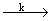 B + n R, n >1 . The second B + n R, n >1 . The second |
| |
reaction is the branching reaction. If the rate law for R is d [R] /dt = k (n-1) [R], how does the concentration of [R] increase with time? |
| |
|
30.4) |
In the three models for oscillatory reactions proposed in equations (30.26), (30.27) and (30.28), identify the |
| |
autocatalytic steps |
| |
|
30.5) |
If one radical produces 3 radicals per second and these three radicals each produce three more radicals in the |
| |
next second and the process continues for 1 minute, how many radicals will be present after 1 minute? Assume that there are no termination reactions. |
| |
|
30.6) |
What is the mechanistic difference between the iodine clock reaction and the "mercury heart " reaction? |
| |
|
30.7) |
What brings about the decrease and the eventual termination of oscillations in concentrations in oscillatory |
| |
reactions? |
| |
|
30.8) |
Distinguish between free radical polymerization and condensation polymerization. |
| |
|
30.9) |
Dacron (polyethylene terephthalate) is formed by the condensation of ethylene glycol with terephthalic acid. Let |
| |
C0 be the initial concentrations of both the acid and the glycol. If the reaction order is one with respect to both the acid and the glycol, using second order kinetics show that the degree of polymerization at time t is given by C0 / C ( t ) = 1/ (1-p ) where p is the fraction of the functional group consumed at time t. |
| |
|
30.10) |
List examples of reactions catalyzed by acids bases and metal ions. Distinguish between homogeneous and |
| |
heterogeneous catalysis. |