Polymerization Reactions. |
| Polymers are molecules that are built from a large number of one or more monomer units. We will consider elementary examples of polymerization, with relatively starightforward mechanisms. Our body too contains a large number of "polymeric systems" such as DNA and proteins but the mechanisms for making these molecules is complex. |
| In chain polymerization, an initiator molecule I interacts with a monomer M to give a reactive monomer unit.
|
| |
I + M 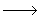 M M + I (initiation) + I (initiation) |
(30.11) |
The reactive M can repeatedly react with other units and propagate the chain through the following propagation steps can repeatedly react with other units and propagate the chain through the following propagation steps |
M + M + M 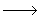 M M 2 2 |
M 2 + M 2 + M 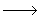 M M 3 3 |
Mn + M 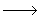 M M n +1 (propagation steps) n +1 (propagation steps) |
(30.12) |
Two large units M K and M K and M l can combine to terminate the sequence of reactions l can combine to terminate the sequence of reactions |
M k + M k + M l l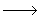 Mk + l ((termination) Mk + l ((termination) |
(30.13) |
| In a polymerization reaction, chains of varying lengths are formed. The art of polymer synthesis is to restrict the growth to a narrow range of desired chain lengths. |
| |
To get an approximate rate law let us make a few assumptions. 1) The total radical concentration R is constant during a major part of the reaction wherein we can apply the steady state approximation, 2) The rate of propagation kP is the same for all n, M n + M n + M 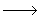 M M n + 1. n + 1. |
3) The rate of termination kT does not depend on chain length M k and M k and M l and 4) there are no side reactions leading to chain truncation, transfer or branching. l and 4) there are no side reactions leading to chain truncation, transfer or branching. |
| |
| If kI is the initiation rate, the rate of formation of R is given by |
| d [ R ] / dt = kI [ M ] [ I ] - 2 k T [ R ] 2 |
(30.14) |
| In the steady state approximation, d [ R ] / dt = 0 which leads to [ R ] = { kI [ M ] [ I ] / 2 kT} 1/2 |
(30.15) |
| The rate of propagation of chains is the rate at which the monomer is consumed, and is given by |
| d [ M ] /dt = - kp [ R ] [ M ] |
(30.16) |
| Substituting the value of R, we have |
| d [ M ] / dt = - kp ( kI / 2 K T ) 1 / 2 [ I ] 1 / 2 [ M ] 3 / 2 |
(30.17) |
| |
| |