The gaseous reaction between H2 and Br2 to give HBr is known to have a rate law |
| d[HBr ] / dt = k [ H2] [ Br2] 3/2 / { [ Br2 ] + k ' { HBr] }. The mechanism that rationalizes the above rate law is as follows. |
| |
|
| 1) Br2 + M 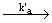 2 Br 2 Br + M (initiation) + M (initiation)
|
| |
2) Br + H2 + H2
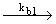 HBr + H HBr + H (branching) (branching) |
| |
3) H + Br2 + Br2 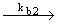 HBr + Br HBr + Br (propagation) (propagation) |
| |
4) H + HBr + HBr
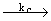 H2 + Br H2 + Br (inhibition) (inhibition) |
5) Br + Br + Br + M + M
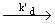 Br2 + M (termination) Br2 + M (termination) |
(30.2) |
| |
| In reaction (1), M is an inert molecule which assists in the process through collisions. Its concentration remains constant during the reaction. |
| In step (1), d [ Br] / dt = 2 k'a [ Br2] [ M ] = 2 ka [ Br2] |
(30.3) |
| |
| where [ M ] has been incorporate into ka through ka = ka' [ M] |
|
| For steps 2 ) and 3) |
| d [ HBr ] / dt = kb1 [ Br] [H2] |
(30.4) |
| d [ HBr ] / dt = kb 2 [ H] [Br2] |
(30.5) |
| For reaction (4), d [ HBr ] /dt = - kc [H] [HBr] |
(30.6) |
| For the termination reaction, |
| d [ Br ] / dt = 2 k'd [ M ] [ Br] 2 = -2 kd [Br]2 |
(30.7) |
| |
If we know the values of the rate constants, we can numerically study the time evolution of all the concentrations on the computer. The steady state approximation gives an approximate and quick solution setting d [ H  ] / dt = d [ Br ] / dt = d [ Br  ] / dt = 0 in the steady state approximation, ] / dt = 0 in the steady state approximation, |
| |
d [ H  ] / dt = 0 = kb1 [ Br] [H2] - kb 2 [ H] [Br2] - kc [ H] [HBr] ] / dt = 0 = kb1 [ Br] [H2] - kb 2 [ H] [Br2] - kc [ H] [HBr] |
d [ Br  ] / dt = 0 = 2ka [ Br2] - kb1 [ Br] [H2] + kb2 [ H] [ Br2] + kc[ H] [Br] - 2 k [ Br]2 ] / dt = 0 = 2ka [ Br2] - kb1 [ Br] [H2] + kb2 [ H] [ Br2] + kc[ H] [Br] - 2 k [ Br]2 |
(30.8) |
| |
| For the rate of production of HBr, we have, using equations for steps 2,3 and 4, |
| d [ HBr] / dt = kb1 [ Br] [ H2] + kb2 [H] [Br2] - kc [H] [HBr] |
(30.9) |
| |
Substituting the steady state concentrations of H  and Br and Br  , we get , we get |
| d [ HBr] / dt = 2 kb1 (ka / kd) 1/ 2 [H2] [Br2] 3 / 2 / { [ Br2 ] + (kc / kb2) [ HBr] } |
(30.10) |
| |
| As mentioned in the beginning, the initiation step of chain reactions could be either thermal or photochemical. Electric sparks are very good initiators. Propagation steps are usually rapid. The concentration of M can be used to control the process. |
| |