| In a chain reaction, a reactive intermediate or a "chain carrier" produced in one step of the reaction takes part in a subsequent step. These chain carriers are repeatedly generated during the reaction untill they are consumed fully. Typical steps in these reactions are initiation, propagation, branching, inhibition and termination. The chain carriers are usually free radicals which are molecules or fragments with one or more unpaired electrons. We will illustrate the mechanism of chain reactions with two examples. |
| a) |
Decomposition of dimethyl ether (methoxymethane) in the gas phase |
| 1) |
CH3OCH 3 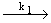 CH3O CH3O + CH3 (initiation) + CH3 (initiation) |
| 2) |
CH3 + CH3OCH3 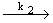 CH4 + CH2OCH3 (branching) CH4 + CH2OCH3 (branching) |
| 3) |
 CH2OCH3 CH2OCH3 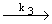  HCHO + CH HCHO + CH 3 (propagation) 3 (propagation) |
| 4) |
 CH3 + CH3 +  CH3 CH3 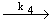 C2H6 (termination) (30.1) C2H6 (termination) (30.1) |
| 5) |
 CH2OCH3 + CH2OCH3 +  CH2OCH3 CH2OCH3 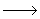 CH3OCH2CH2OCH3 (termination) CH3OCH2CH2OCH3 (termination) |
| 6) |
CH3 + +  CH2OCH3 CH2OCH3 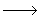 CH3CH2OCH3 (termination) CH3CH2OCH3 (termination) |
| Among these reactions, not all steps are dominant. If the termination step (step 4) is dominant, it can be shown that the order of the reaction with respect to the reactant is 1.5. |