Measurement of pK |
| In the Bronsted Lowry classification scheme, an acid is a proton donor and a base is a proton acceptor. The dissociation equilibrium of acid HA in water may be written as |
|
|
HA (aq) + H2O (l) 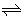 H3O+ (aq) + A- (aq) H3O+ (aq) + A- (aq) |
| The equilibrium constant (in this case, an ionization constant) Ka is defined as |
| Ka = {aH3O+ aA- / aHA} aq |
| The activity of water, aH3O is taken to be unity. The value of pKa is defined as |
| pKa = - log10 Ka |
| A- is called the conjugate base of acid HA. |
| For a base B which is a proton acceptor, BH+ is the conjugate acid and equilibria as above can be easily written. For diprotic acids, we can define two equilibria, with the first ionization constant Ka1 and a second ionization constant Ka2. pKa values measure the strengths of acids . The pKa2 value of H2SO4 is 1.925. Smaller the pKa, stronger the acid. |
| |
| In the acid base titration of a strong acid with strong base, the end point has a pH of 7, at 25oC. For the titration of strong acid with weak base or weak acid with strong base the end point lies at different pH values (other than 7) due to solvolysis (association of solvent molecules with ionic or other species). We need to know the pH of the solution at each stage of titration of say weak acid with strong base to determine the pKa of the acid. We need to consider the following three criteria. |
| 1) The two equilibria associated with the dissociation of a weak acid are, |
HA(aq) + H2O (l) 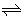 A- (aq) + H3O+ (aq) , and A- (aq) + H3O+ (aq) , and |
2H2O (l) 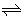 OH- (aq) + H3O+ (aq) OH- (aq) + H3O+ (aq) |
| Ka = [H+] [A-] / [HA], Kw = [H+] [OH-] |
| |
| 2) The overall electroneutrality of the solution, |
| [H+] + [M+] = [A-] + [OH-] |
| Here MOH is the strong base (fully dissociating) and [M+] = amount of base added = [MOH] added, and |
| |