| Introduction |
By measuring the emf of suitable galvanic cells, it is possible to calculate the changes in the thermodynamic functions  G, G,  H and H and  S, the solubility product, the pH of a solution, activities of electrolytes, and pKa and pKb. In analytical chemistry too, emf measurements have wide ranging applications. S, the solubility product, the pH of a solution, activities of electrolytes, and pKa and pKb. In analytical chemistry too, emf measurements have wide ranging applications. |
| |
 G and G and  S are determined from emf measurements and S are determined from emf measurements and  H is evaluated from the Gibbs - Helmohltz equation. Emf measurements are normally made at constant temperature. Since emf is affected by changes in temperature, the temperature coefficient of emf viz., d H is evaluated from the Gibbs - Helmohltz equation. Emf measurements are normally made at constant temperature. Since emf is affected by changes in temperature, the temperature coefficient of emf viz., d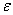 /dT can be determined by measuring the emf at 2 or 3 different temperatures. It is related to /dT can be determined by measuring the emf at 2 or 3 different temperatures. It is related to  S as shown below. Differentiating S as shown below. Differentiating 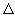 G = -nF G = -nF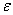 w.r.t. temperature (and replacing w.r.t. temperature (and replacing 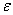 by E for convenience ), by E for convenience ), |
|
d ( G) /dT = - nF (dE /dT) G) /dT = - nF (dE /dT) |
| since d( G) /dT = - G) /dT = -  S we have S we have
|
| |
 S = nF [dE /dT] S = nF [dE /dT] |
(24.1) |
 H at any given temperature can be either calculated from the Gibbs-Helmholtz equation or from the following standard relation for free energy. H at any given temperature can be either calculated from the Gibbs-Helmholtz equation or from the following standard relation for free energy. |
| |
 G = G =  H - T H - T S S |
(24.2) |
| |
| Example 24.1: |
| The galvanic cell |
Ag(s) | AgCl (s) | KCl ( 1.0 M ) I Hg2 Cl2 (s) | Hg ( l ) |
| is an example of cell without a liquid junction since both the electrodes dip into the same electrolyte. The emf of the cell is 0.058V at 298 K and 0.0614V at 308 K. |
The electrode reaction are |
(i) RHS electrode: Hg2Cl2 + 2e - 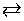 2Hg ( l ) + 2Cl - 2Hg ( l ) + 2Cl - |
LHS electrode: 2Ag (s) + 2Cl - 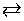 2AgCl+ 2e 2AgCl+ 2e |
The cell reaction is 2Ag (s ) + Hg2Cl2 (s) 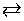 2Hg ( 1 ) + 2AgCl ( s ) 2Hg ( 1 ) + 2AgCl ( s ) |
| The thermodynamic function are calculated as shown below. |
(ii)  G o = -nFEo =-2 x 96500C x 0.058 V = -11194J mol -1 G o = -nFEo =-2 x 96500C x 0.058 V = -11194J mol -1 |
| |
(iii) dEo /dT 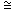  Eo / Eo /  T = ( 0.0614 - 0.058) / 10 = 3.40 x 10 -4 VK -1 T = ( 0.0614 - 0.058) / 10 = 3.40 x 10 -4 VK -1 |
| |
(iv)  So = nF dEo / dT = 2 x 965000C x 3.4 x 10 -4 VK -1 = 57.9 JK -1 mol -1 So = nF dEo / dT = 2 x 965000C x 3.4 x 10 -4 VK -1 = 57.9 JK -1 mol -1 |
| |
(v)  Go = Go =  Ho - T Ho - T So So |
 Ho = Ho =  Go + T Go + T So So |
= -11194 + 298 x 57.9 = 6060 J mol -1 |
| These details have already been described in the earlier lecture 22 and are given here for completeness and continuity. |
| |