The solubility product Ksp (ABn 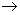 An+ + n B - ; Ksp is defined as Ksp= [A n+ ] [B - ] n ) is an equilibrium constant and its value can be predicted from a knowledge of the standard electrode potentials. The electrodes are chosen in such a way , that the overall cell reaction is the solubility product equilibrium. A suitable galvanic cell is thus set up and its emf is determined experimentally. Even if measurements can not be made, it is possible to arrive at the approximate values of Ksp from appropriate half cell reaction potentials as shown below. An+ + n B - ; Ksp is defined as Ksp= [A n+ ] [B - ] n ) is an equilibrium constant and its value can be predicted from a knowledge of the standard electrode potentials. The electrodes are chosen in such a way , that the overall cell reaction is the solubility product equilibrium. A suitable galvanic cell is thus set up and its emf is determined experimentally. Even if measurements can not be made, it is possible to arrive at the approximate values of Ksp from appropriate half cell reaction potentials as shown below. |