Let f + and f - be the scattering factors of Na+ and Cl - respectively. Just as the atom inside a BCC unit cell contributes one atom to the unit cell, an atom inside the unit cell contributes a strength of f to the scattered intensity. An atom shared between faces contributes f / 2, an atom at a corner contributes f / 8 and an atom on the edge of a side contributes f / 4 . We will also need the two useful relations given below |
|
e 0 = e 2
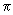 i n = 1, e i i n = 1, e i 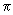 = -1, (e i = -1, (e i
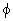 + e - i + e - i 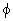 ) / 2 = cos ) / 2 = cos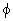 |
(19.9) |
where n is an integer |
| There are 27 atoms in the unit cell of fig 19.4. Summing over all ions, |
| |
| F h k l = f +[ 1/8 +1/8 e 2 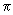 i l +...........1/ 2 e 2 i l +...........1/ 2 e 2 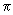 i (1 / 2 h +1 / 2 k + l ) ] + f - [ e2 i (1 / 2 h +1 / 2 k + l ) ] + f - [ e2 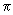 i (1 / 2 h +1 / 2 k + 1 / 2l ) + 1/4 e 2 i (1 / 2 h +1 / 2 k + 1 / 2l ) + 1/4 e 2 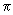 i ( h / 2) ....... ................................. + 1/4 e 2 i ( h / 2) ....... ................................. + 1/4 e 2 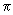 i(1 / 2 h +1) ] i(1 / 2 h +1) ]
|
(19.10) |
| Using Eq (19.9), Eq (19.10) can be simplified to |
| |
F h k l = f + [ 1 + cos ( h + k )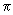 + cos ( h + l ) + cos ( h + l )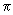 + cos (k + l) + cos (k + l)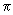 ] + f - [ (-1) h + k + l + cos k ] + f - [ (-1) h + k + l + cos k 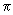 + cos l + cos l 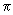 + cos h + cos h 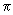 ] ] |
(19.11) |
| |
since cos n 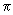 = ( -1)n, we get = ( -1)n, we get |
| |
| Fh k l = f + [ 1 + (-1) h + k + (-1) h + l + (-1) l + k ] + f - [ (-1) h + k + l + (-1)h + (-1) k + (-1) l ] |
(19.12) |
| |
| We can easily deduce from Eq (19.12) that |
| |
|
| Fh k l = f + (1+ 1+ 1 + 1 ) + f - (1+ 1+ 1 + 1 ) = 4 f + + 4 f - |
(19.13) |
| if all h, k and l are even |
| |
| Fh k l = 4 ( f + - f - ) if each of h, k and l is odd |
(19.14) |
| and |
| |
| Fh k l = 0 if one is odd and two are even or two are odd and one is even |
(19.15) |
| |
| This analysis now justifies our assertion made in Fig 19.12. This also brings out the fact that even if two unit cells are the same (e.g., FCC or BCC ), their diffraction patterns can be very different if f + and f - in Eqs (19.13) to (19.15) are different. This is illustrated in Fig 19.5. |
| |