In addition to X - rays, neutrons and electrons can also be used in diffraction techniques by carefully choosing their wavelengths to lie between 10 pm to 200 pm. The wavelength
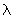 of any object may be obtained from the de Broglie relation of any object may be obtained from the de Broglie relation
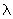 = h / p where h is Planck's constant and p, the momentum of the particle. = h / p where h is Planck's constant and p, the momentum of the particle.
|