Continuing our discussion from Fig 19.2, the total amplitude at the detector from layers of atoms A and layers of atom B is |
|
F h k l = f A + f B e i
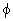 h k l h k l |
(19.6) |
where
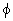 is the phase difference between the h k l reflections from A atoms and B atoms. is the phase difference between the h k l reflections from A atoms and B atoms. |
| The intensity ( I ) of X-rays is proportional to the square modulus of the amplitude given by Eq (19.6). We get |
|
|
I
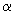 F * h k l F h k l = ( f A + f B e - i F * h k l F h k l = ( f A + f B e - i 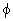 h k l ) ( f A + f B e i h k l ) ( f A + f B e i 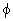 h k l) h k l)
|
= fA 2 + f B 2 + 2 f A f B cos 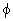 h k l h k l |
(19.7) |
| |
We thus observe that depending on 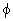 h k l ( ie the phase difference between the h, k, l planes expressed in terms of the distance between layers A and B and fractions x, y and z ) the intensities of two lattices containing different sets of atoms is different. If the unit cell contains several types of atoms labeled by j with scattering factors fj and coordinates (x j a, y j b, z j c) then the total amplitude of the wave scattered by the h k l planes is h k l ( ie the phase difference between the h, k, l planes expressed in terms of the distance between layers A and B and fractions x, y and z ) the intensities of two lattices containing different sets of atoms is different. If the unit cell contains several types of atoms labeled by j with scattering factors fj and coordinates (x j a, y j b, z j c) then the total amplitude of the wave scattered by the h k l planes is |
| |
F h k l =
 f j e i f j e i 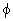 h k l ( j ) h k l ( j ) |
(19.8) |
where 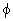 h k l ( j ) = 2 h k l ( j ) = 2
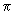 (h x j + k y j + l z j) and (h x j + k y j + l z j) and |
| the summation is over all the atoms in the unit cell. F h k l is known as the structure factor and the intensity of reflection from the h k l plane is proportional to | Fh k l | 2 |
| Let us use Eq (19.8) to analyze the structure factor of NaCl, whose structure is shown in fig 19.4. The coordinates of the atoms are also shown as (0, 0, 0), (1 1 1), (1/2, 1/2, 0) and so on. |
| |
|
| |
Figure 19.4 NaCl structure with coordinates. |
| |