We have seen that Eq (19.3) gives the scattering factor of an atom or an ion in terms of its electron density. This scattering factor is a function of the wave - vector
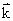 , which gives the vector in the direction of the propagation of the wave. The magnitude of this vector is k. From an analysis of Eq (19.3) we find that fA is greatest in the forward direction and smaller in other directions. A sketch of fA as a function of k is given in Fig 19.3. , which gives the vector in the direction of the propagation of the wave. The magnitude of this vector is k. From an analysis of Eq (19.3) we find that fA is greatest in the forward direction and smaller in other directions. A sketch of fA as a function of k is given in Fig 19.3.
|