For a BCC lattice, x = y = z = 1/ 2 since an atom is present at x = y = z = 1/ 2 a where a = edge length. In such a situation, the phase difference between the A and B layers is ( h + k+ l )
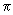 . This means that when the value of h + k + l = odd, there is destructive interference and all reflections with h + k + l = odd are absent in a BCC lattice. Our analysis so far has assumed identical atoms in the A and B layers. . This means that when the value of h + k + l = odd, there is destructive interference and all reflections with h + k + l = odd are absent in a BCC lattice. Our analysis so far has assumed identical atoms in the A and B layers.
|