UV-VIS Spectroscopy:
As the absorbance of a sample is proportional to the number of absorbing molecules in the spectrometer light beam (e.g. their molar concentration in the sample tube), it is necessary to correct the absorbance value for this and other operational factors if the spectra of different compounds are to be compared in a meaningful way. The corrected absorption value is called "molar absorptivity", and is particularly useful when comparing the spectra of different compounds and determining the relative strength of light absorbing functions (chromophores). The molar absorptivity (ε) is defined as:

where A is absorbance, c is sample concentration in moles/liter and l is the length of light path through the sample in cm.
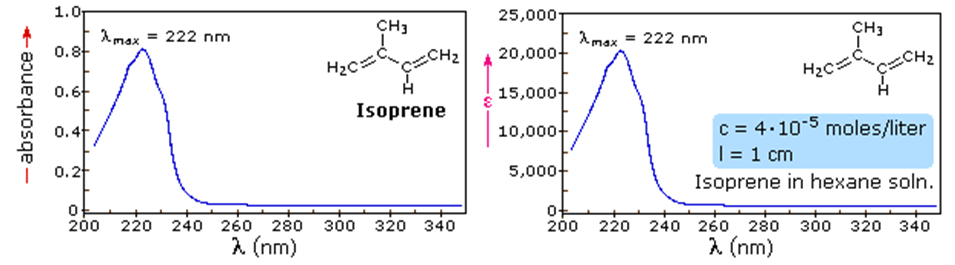
Figure 37.07: Absorbance spectrum and molar absorptivity of Isoprene in hexane solution [1].
Ref.[1]. http://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/spectrpy/uv-vis/spectrum.htm.