UV-VIS Spectroscopy:
(2) The photon energy must be equal to the difference in the energy between the sample's quantized energy states. Figure 37.02 shows a simplified view of a photon's absorption to change the energy state of the materials from ground state to excited state. However, this schematic picture does not provide the information about what types of energy states are involved, which transitions between energy states are likely to occur, and the appearance of the resulting spectrum.
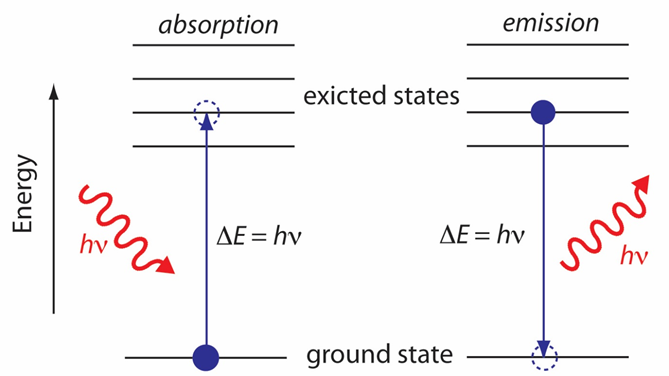
Figure 37.02 : Simplified energy diagram showing the absorption and emission of a photon by an atom or a molecule.