UV-VIS Spectroscopy:
Figure 37.07 displays the absorbance and molar absorptivity and absorbance of an isoprene spectrum. The entire vertical absorbance scale can be changed to a molar absorptivity once the information about the samples as given earlier equation is known.
UV-VIS spectrum also helps to understand the importance of conjugation. For example, the spectrum shown left of Figure 37.08 illustrates that conjugation of double and triple bonds, which shift the absorption maximum to longer wavelengths. From the polyene spectra displayed in the right diagram, it is clear that each additional double bond in the conjugated pi-electron system shifts the absorption maximum about 30 nm in the same direction.
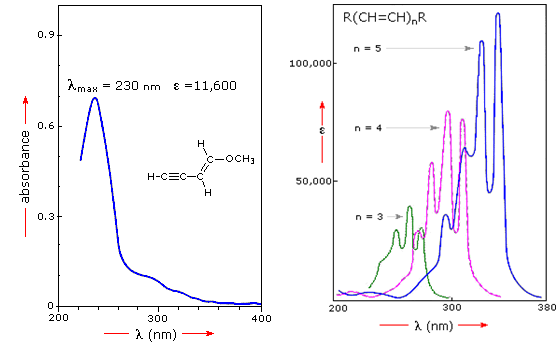
Figure 37.08: Absorbance spectrum of conjugation [1].
This fine structure reflects not only the different conformations such systems may assume, but also electronic transitions between the different vibrational energy levels possible for each electronic state.
Vibrational fine structure of this kind is most pronounced in vapor phase spectra, and is increasingly broadened and obscured in solution.
Quiz 37:
(Q37.1) What are the phenomena that occur when the light interacts with materials?
(Q37.2) What are the conditions to be satisfied for absorption?
(Q37.3) What are the possible transition observable for Cl atom?
(Q37.4) What is the advantage of double-beam spectrophotometers over single-beam spectrophotometers?