UV-VIS Spectroscopy:
As listed in Table 1, the energy of UV-VIS electromagnetic radiation is sufficient to cause a change in an atom's valence electron configuration. Let us take an example to understand the process. Consider the sodium (Na) atom, which has a single valence electron [(1s)2 (2s)2 (2p)6 (3s)1] in its 3s orbital as shown in Figure 37.03.
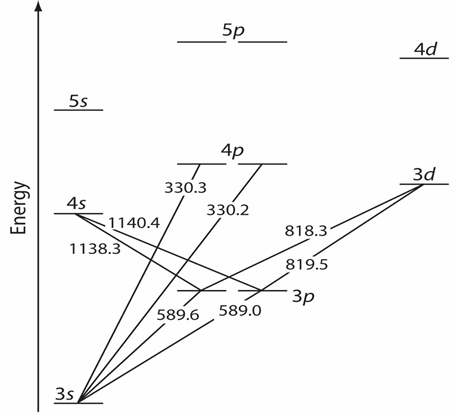
Figure 37.03: Valence shell energy level diagram for sodium. The wavelengths corresponding to several transitions are shown.
When Na absorbs a photon, then electron in 3s orbital is excited from a lower-energy atomic orbital to an orbital of higher energy.
While it is clearly understood that not all possible transitions between atomic orbitals are allowed, the only allowed transitions are those in which there is a change of ± 1 in the orbital quantum number (l); thus transitions from s → p orbitals are allowed, and transitions from s → d orbitals stand forbidden.
When light passes through a sample, its power decreases as some of it is absorbed. This is often described quantitatively by two separate, but related terms: transmittance and absorbance. Figure 37.04 displays schematic view of light passing through the sample kept in a container and blank container.