Phase Space
If the position and momentum of each atom of a gas in an enclosure is to be determined, we require six quantities, e.g., x, y, z, px, py and pz. A six dimensional hyperspace with three location and three momentum coordinates is known as a phase space. Each particle in space can be represented as a point in a phase space and it is termed as phase point.
Let us subdivide the phase space into small elements of volume, called cells, and volume of one cell, H, is:
H = dx.dy.dz.dpx.dpy.dpz |
(5.13) |
This volume is small compared to the dimensions of the system, but large enough to contain many atoms. Every atom of the gas must be in a cell. The cells are numbered 1, 2, 3, ….., I, … and the number of particles in the cells are N1, N2, N3,.........., Ni,....... with Ni > 1. The basic problem of statistical mechanics is to determine how the particles distribute themselves in the cells of phase space.
According to the Heisenberg's uncertainity principle, ΔxΔpx = h, ΔyΔpy = h, ΔzΔpz = h. The total uncertainity in locating a particle in phase space is
Δx.Δy.Δz.Δpx.Δpy.Δpz = h3 |
(5.14) |
The particle lies somewhere within an element of phase space of volume h3. This is known as a compartment. The number of compartment per cell is
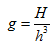 |
(5.15) |
where g >> 1 and h3 has the dimension of volume in phase space.
h3 = (length)3 X (momentum)3 |
(5.16) |
A quantum state corresponds to a volume h3 in space, and an energy level corresponds to a cell of volume H, so g is nothing but the degeneracy of the energy level.