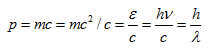Wave particle duality
de Broglie equation ![]()
de Broglie equation relates the momentum of the photon ( or electron) with its wave length, representing the wave-particle duality. The total energy ε and mass of a moving particle m are related by Einstein's famous equation,
E = mc2 |
(5.5) |
where c is the velocity of light ( 3 X 108 m/s). For a photon ( photon does not have mass but has momentum) of electromagnetic radiation, Planck's equation gives,
ε = hv |
(5.6) |
where h is the Planck's constant and v is the frequency of the wave.
Now momentum p of the photon is
|
(5.7) |
When this equation is applied to a heavy particle, the wavelength is so small that the modification made on rectilinear motion by the wave can be neglected, thus reverting back to classical mechanics.