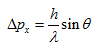Heisenberg's Uncertainity Principle (The quantitative limit on the product of the uncertainities in position and momentum of a particle.)
Suppose we want to determine the position and momentum of a particle, say an electron. The position of the electron can be determined by illuminating it with a light source of wavelength λ and observing through a microscope. The uncertainity Δx of the position in x-direction is known to be a function of λ and the aperature of the lens, given by the angle Θ, or
|
(5.8) |
The uncertainiy Δx can be made small by selecting radiation of short wave length, such as X-rays and γ- rays. But the photon of short wave length or high frequency striking the electron yields extensive scattering. The change of momentum of the electron can be found from the angle of scattering of the photon. The angle must be within the visible range of the lens. The uncertainity of x-momentum is given by
|
(5.9) |
where h is the Planck's constant. The product of uncertainities in position and momentum is
Δx.Δpx = h |
(5.10) |
This is known as Heisenberge's uncertainity principle.
Similarly,
Δy.Δpy = h |
(5.11) |
and
Δz.Δpz = h |
(5.12) |
Attempt to improve the accuracy or to decrease the uncertainity for determination of momentum or position results in a corresponding decrease in the accuracy of the conjugate variable.