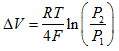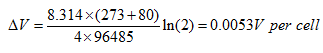Practical fuel cell voltages
In practice the actual cell voltage is less than this. Now of course this applies to ordinary batteries too, as when current is drawn out of any electric cell the voltage falls, due to internal resistances. However, with a fuel cell this effect is more marked than with almost all types of conventional cell. Figure 6 shows a typical voltage/current density curve for a good PEM fuel cell. It can be seen that the voltage is always less, and is often much less, than the 1.18V that would be obtained if all of the Gibbs energy were converted into electrical energy.
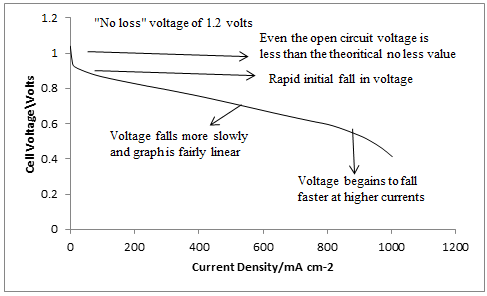
Fig. 6 Graph showing the voltage from a typical good quality PEM fuel cell operating on air at about 80°C
There are three main reasons for this loss of voltage, as detailed below.
• The energy required to drive the reactions at the electrodes, usually called the activation energy, causes a voltage drop. This is especially a problem at the air cathode, and shows itself as a fairly constant voltage drop. This explains the initial fall in voltage even at quite low currents.
• The resistance of the electrolyte and the electrodes causes a voltage drop that more or less follows Ohm's law, and causes the steady fall in voltage over the range of currents. This is usually called the Ohmic voltage loss.
• At very high currents, the air gets depleted of oxygen, and the remnant nitrogen gets in the way of supplying fresh oxygen. This result is a fall in voltage, as the electrodes are short of reactant. This problem causes the more rapid fall in voltage at higher currents, and is called mass transfer or concentration voltage loss.
The effect of pressure and gas concentration
The values for the changes in the Gibbs free energy given in Tables II and III all concern pure hydrogen and oxygen, at standard pressure, 100 kPa. However, as well as changing with temperature, as shown in these tables, the Gibbs energy changes with pressure and concentration.
A full treatment of these issues is beyond a book such as this, and it can easily be found elsewhere. Suffice to say that the relationship is given by a very important fuel cell equation derived from the work of Nernst. It can be expressed in many different forms, depending on what issue is to be analyzed. For example, if the change of system pressure is the issue, then the Nernst equation takes the form:
|
(18) |
Where ΔV is the voltage increase if the pressure changes from P 1 to P 2 . Other causes of voltage change are a reduction in voltage caused by using air instead of pure oxygen. The use of hydrogen fuel that is mixed with carbon dioxide, as is obtained from the ‘reforming' of fuels such as petrol, methanol or methane, also causes a small reduction in voltage.
For high temperature fuel cells the Nernst equation predicts very well the voltage changes. However, with lower temperature cells, such as are used in electric vehicles, the changes are nearly always considerably greater than the Nernst equation predicts. This is because the ‘activation voltage drop' mentioned in the last section is also quite strongly affected by issues such as gas concentration and pressure. This is especially the case at the air cathode.
The voltage increase resulting from a doubling of the system pressure would be:
|
(19) |
However, in practice the voltage increase would typically be about 0.04 V, nearly ten times as much. Even so, we should note that the increase is still not large, and that there is considerable energy cost in running the system at higher pressure. Indeed, it is shown elsewhere that the energy gained from a higher voltage is very unlikely to be greater than the energy loss in pumping the air to higher pressure.