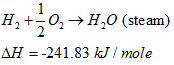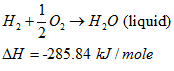However, the energy of the chemical inputs and output is not so easily defined. At a simple level we could say that it is the chemical energy of the H2 , O2 and H2 O that is in question. The problem is that chemical energy is not simply defined, and terms such as enthalpy, Helmholtz function and Gibbs free energy are used. In recent years the useful term ‘energy' has become quite widely used, and the concept is particularly useful in high temperature fuel cells, though we are not concerned with these here. There are also older (but still useful) terms such as calorific value.
In the case of fuel cells it is the Gibbs free energy that is important. This can be defined as the energy available to do external work, neglecting any work done by changes in pressure and/or volume. In a fuel cell the external work involves moving electrons round an external circuit; any work done by a change in volume between the input and output is not harnessed by the fuel cell. Energy is all the external work that can be extracted, including that due to volume and pressure changes. Enthalpy, simply put, is the Gibbs free energy plus the energy connected with the entropy. The enthalpy H , Gibbs free energy G and entropy S are connected by the well-known equation:
G = H - TS |
(5) |
The energy that is released by a fuel cell is the change in Gibbs energy before and after a reaction, so the energy released can be represented by the equation:
ΔG = Goutputs - Ginputs |
(6) |
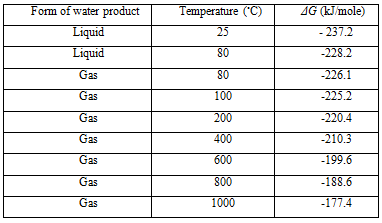
However, the Gibbs free energy change is not constant, but changes with temperature and state (liquid or gas). Table II below shows ΔG for the basic hydrogen fuel cell reaction for a number of different conditions. Note that the values are negative, which means that energy is released.
|
(7) |
Table II ΔG for the reaction H2 + 12 O2 → H2O at various temperatures
If there are no losses in the fuel cell, or as we should more properly say, if the process is reversible, then all this Gibbs free energy is converted into electrical energy. We could thus define the efficiency of a fuel cell as:
|
(8) |
Since a fuel cell uses materials that are usually burnt to release their energy, it would make sense to compare the electrical energy produced with the heat that would be produced by burning the fuel. This is sometimes called the calorific value, though a more precise description is the change in enthalpy of formation. Its symbol is ΔH. As with the Gibbs free energy, the convention is that ΔH is negative when energy is released. So to get a good comparison with other fuel using technologies, the efficiency of the fuel cell is usually defined as:
|
(9) |
However, even this is not without its ambiguities, as there are two different values that we can use for ΔH. For the burning of hydrogen:
|
(10) |
whereas if the product water is condensed back to liquid, the reaction is:
|
(11) |
The difference between these two values for ΔH (44.01 kJ/mole) is the molar enthalpy of vaporization of water. The higher figure is called the higher heating value (HHV), and the lower, quite logically, the lower heating value (LHV). Any statement of efficiency should say whether it relates to the higher or lower heating value. If this information is not given, the LHV has probably been used, since this will give a higher efficiency figure.
We can now see that there is a limit to the efficiency, if we define it as in Eq. 4. The maximum electrical energy available is equal to the change in Gibbs free energy, so:
|
(12) |
This maximum efficiency limit is sometimes known as the thermodynamic efficiency. Table III gives the values of the efficiency limit, relative to the higher heating value, for a hydrogen fuel cell. The maximum voltage obtainable from a single cell is also given.
The graphs in Fig. 5 show how these values vary with temperature, and how they compare with the Carnot limit, which is given by the equation:
|
(13) |