In this lecture the energy storage (fuel cell) is presented. The following topics are covered in this lecture:
• Fuel cell
• Issues in fuel cell
• Hydrogen fuel cell
• Fuel cell thermodynamics
• Main reasons for loss in voltage
Fuel Cell
Introduction
Fuel cells are hardly a new idea. They were invented in about 1840, but they are yet to really make their mark as a power source for electric vehicles. However, this might be set to change over the next 20 or 30 years. Certainly most of the major motor companies are spending very large sums of money developing fuel cell powered vehicles. The basic principle of the fuel cell is that it uses hydrogen fuel to produce electricity in a battery-like device to be explained in the next section. The basic chemical reaction is:
|
(1) |
The product is thus water, and energy. Because the types of fuel cell likely to be used in vehicles work at quite modest temperatures (~ 85°C) there is no nitrous oxide produced by reactions between the components of the air used in the cell. A fuel cell vehicle could thus be described as zero-emission. Furthermore, because they run off a fairly normal chemical fuel (hydrogen), very reasonable energies can be stored, and the range of fuel cell vehicles is potentially quite satisfactory. They thus offer the only real prospect of a silent zero-emission vehicle with a range and performance broadly comparable with IC engine vehicles. It is not surprising then that there have, for many years, been those who have seen fuel cells as a technology that shows great promise, and could even make serious inroads into the domination of the internal combustion engine.
Main issues in the fuel cell
There are many problems and challenges for fuel cells to overcome before they become a commercial reality as a vehicle power source. The main problems centre on the following issues.
• Cost: Fuel cells are currently far more expensive than IC engines, and even hybrid IC/electric systems.
• Water management: It is not at all self-evident why water management should be such an important and difficult issue with automotive fuel cells.
• Cooling: The thermal management of fuel cells is actually rather more difficult than for IC engines.
• Hydrogen supply: Hydrogen is the preferred fuel for fuel cells, but hydrogen is very difficult to store and transport. There is also the vital question of ‘where does the hydrogen come from' these issues are so difficult and important, with so many rival solutions.
However, there is great hope that these problems can be overcome, and fuel cells can be the basis of less environmentally damaging transport.
Hydrogen Fuel Cells: Basic Principles
Electrode reactions
We have seen that the basic principle of the fuel cell is the release of energy following a chemical reaction between hydrogen and oxygen. The key difference between this and simply burning the gas is that the energy is released as an electric current, rather that heat. How is this electric current produced?
To understand this we need to consider the separate reactions taking place at each electrode. These important details vary for different types of fuel cell, but if we start with a cell based on an acid electrolyte, we shall consider the simplest and the most common type.
At the anode of an acid electrolyte fuel cell the hydrogen gas ionizes, releasing electrons and creating H+ ions (or protons).
|
(2) |
This reaction releases energy. At the cathode, oxygen reacts with electrons taken from the electrode, and H+ ions from the electrolyte, to form water.
|
(3) |
Clearly, for both these reactions to proceed continuously, electrons produced at the anode must pass through an electrical circuit to the cathode. Also, H+ ions must pass through the electrolyte. An acid is a fluid with free H+ ions, and so serves this purpose very well. Certain polymers can also be made to contain mobile H+ ions.
Different electrolytes
The reactions given above may seem simple enough, but they do not proceed rapidly in normal circumstances. Also, the fact that hydrogen has to be used as a fuel is a disadvantage. To solve these and other problems many different fuel cell types have been tried. The different types are usually distinguished by the electrolyte that is used, though there are always other important differences as well.
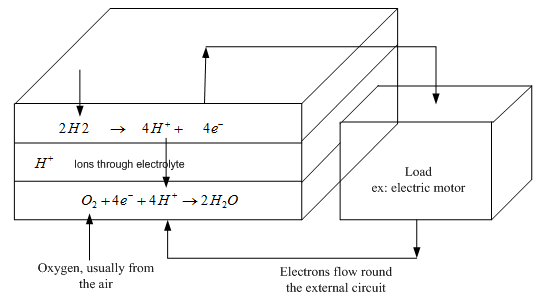
Fig. 1 The reactions at the electrodes, and the electron movement, in a fuel cell with an acid Electrolyte
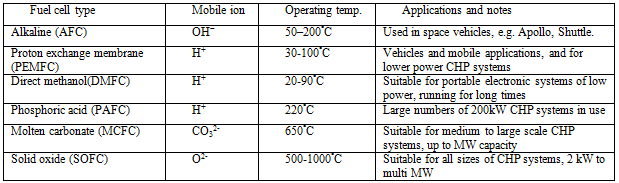
Table I Data for different types of fuel cell