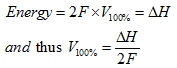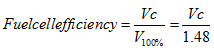whereT1 is the higher temperature, and T2 the lower, of the heat engine. The graph makes clear that the efficiency limit of the fuel cell is certainly not 100%, as some supporters of fuel cells occasionally claim. Indeed, above the 750 ◦°C the efficiency limit of the hydrogen fuel cell is actually less than for a heat engine. Nevertheless, the PEM fuel cells used in vehicles operate at about 80°C, and so their theoretical maximum efficiency is actually much better than for an IC engine.
Efficiency and the fuel cell voltage
A very useful feature of fuel cells is that their efficiency can be very easily found from their operating voltage. The reasoning behind this is as follows. If one mole of fuel is reacted in the cell, then two moles of electrons are pushed round the external circuit;
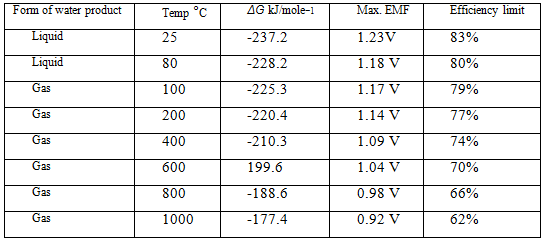
Table III ΔG, maximum EMF, and efficiency limit (HHV) for hydrogen fuel cells
Energy = Charge X Voltage |
(14) |
The Faraday constant F gives the charge on one mole of electrons. So, when one mole of hydrogen fuel is used in a fuel cell, if it were 100% efficient, as defined by Eq. 4, then we would be able to say that:
|
(15) |
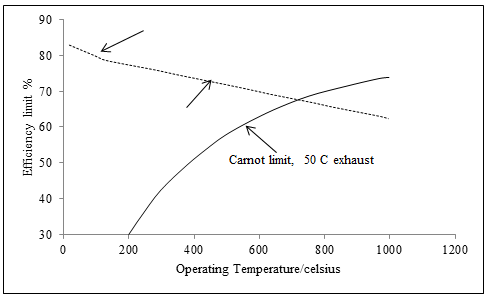
Fig. 5 Maximum hydrogen fuel cell efficiency at standard pressure, with reference to the higher heating value.
The two values for ΔH given above, we can easily calculate that the ‘100% efficient' voltage for a single cell is 1.48V if using the HHV or 1.25V if using the LHV. Now of course a fuel cell never is, and we have shown in the last section never can be, 100% efficient. The actual fuel cell voltage will be a lower value, which we can call Vc . Since voltage and electrical energy are directly proportional, it is clear that
|
(16) |
Clearly it is very easy to measure the voltage of a fuel cell. In the case of a stack of many cells, remember that the voltage of concern is the average voltage one cell, so the system voltage should be divided by the number of cells. The efficiency can thus be found remarkably easily.
It is worth noting in passing that the maximum voltage of a fuel cell occurs when 100% of the Gibbs free energy is converted into electrical energy. Thus we have a ‘sister' equation to Eq. 4, giving the maximum possible fuel cell voltage:
|
(17) |
This is also a very important fuel cell equation, and it was used to find the figures shown in the fourth column of Table III.