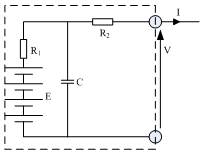
Fig. 1 Example of a more refined equivalent circuit model of a battery. This models some of the dynamic behavior of a battery
| V = E - IR | ...................................................................(2) |
In the case of nickel-based batteries such a simple formula cannot be constructed. The voltage/state of charge curve is far from linear. Fortunately it now very easy to use mathematical software, such as MATLAB, to find polynomial equations that gives a very good fit to the results. One such, produced from experimental results from a NiCad traction battery is:
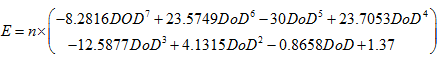 |
..................................(3) |
The purpose of being able to simulate battery behavior is to use the results to predict vehicle performance. In other words we wish to use the result in a larger simulation. This is best done in software such as MATLAB or an EXCEL spreadsheet.
The simple battery model of Figure 1 now has a means of finding E , at least for some battery types. The internal resistance also needs to be found. The value of R is approximately constant for a battery, but it is affected by the state of charge and by temperature. It is also increased by misuse, and this is especially true of lead acid batteries.
Modeling battery capacity
The capacity (10Ahr) of a battery is reduced if the current is drawn more quickly. Drawing 1A for 10 hours does not take the same charge from a battery as running it at 10A for 1 hour. This phenomenon is particularly important for electric vehicles, as in this application the currents are generally higher, with the result that the capacity might be less than is expected. It is important to be able to predict the effect of current on capacity, both when designing vehicles, and when making instruments that measure the charge left in a battery: battery fuel gauges.
The best way to do this is using the Peukert model of battery behavior. Although not very accurate at low currents, for higher currents it models battery behavior well enough. The starting point of this model is that there is a capacity, called the Peukert Capacity, which is constant, and is given by the equation:
| (4) |
where k is a constant (typically about 1.2 for a lead acid battery) called the Peukert Coefficient. Suppose a battery has a nominal capacity of 40 Ah at the 5 h rate. This means that it has a capacity of 40 Ah if discharged at a current of:
| (5) |
If the Peukert Coefficient is 1.2, then the Peukert Capacity is:
| (6) |
We can now use equation 4 (rearranged) to find the time that the battery will last at any current I.
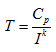 |
(7) |
This is for a nominally 42 Ah battery (10 h rate), and shows how the capacity changes with discharge time. This solid line in Figure 2 shows the data of Figure 1 in a different form, i.e. it shows how the capacity declines with increasing discharge current. Using methods described below, the Peukert Coefficient for this battery has been found to be 1.107. From equation 6 we have: