In this lecture the mathematical modeling for energy storage devices are presented. The following topics are covered in this lecture:
• Battery Charging
• The Designer's Choice of Battery
• Use of Batteries in Hybrid Vehicles
• Battery Modeling
Battery Charging
The battery charging involves:
• A battery charger
• Charge equalization
Battery chargers
Charging a modern vehicle battery is not a simple matter of providing a constant voltage or current through the battery, but requires very careful control of current and voltage. The best approach for the designer is to buy commercial charging equipment from the battery manufacturer or another reputed battery charger manufacturer.
When the vehicle is to be charged in different places where correct charging equipment is not available, the option of a modern light onboard charger should be considered.
Except in the case of photoelectric panels, the energy for recharging a battery will nearly always come from an alternating current (AC) source such as the mains. This will need to be rectified to direct current (DC) for charging the battery.
The rectified DC must have very little ripple, it must be very well ‘smoothed'. This is because at the times when the variation of the DC voltage goes below the battery voltage, no charging will take place, and at the ‘high point' of the ripple it is possible that the voltage could be high enough to damage the battery. The higher the DC current, the harder it is for rectifiers to produce a smooth DC output, which means that the rectifying and smoothing circuits of battery chargers are often quite expensive, especially for high current chargers.
Charge equalization
A problem with all batteries is that when current is drawn not all the individual cells in the battery lose the same amount of charge. Since a battery is a collection of cells connected in series, this may at first seem wrong; after all, exactly the same current flows through them all.
However, it does not occur because of different currents (the electric current is indeed the same) it occurs because the self-discharge effects we have noted in the case of lead acid batteries) take place at different rates in different cells. This is because of manufacturing variations, and also because of changes in temperature; the cells in a battery will not all be at exactly the same temperature. The result is that if nominally 50% of the charge is taken from a battery, then some cells will have lost only a little more than this, say 52%, while some may have lost considerably more, says 60%. If the battery is recharged with enough for the good cell, then the cells more prone to self-discharge will not be fully re-charged. The effect of doing this repeatedly is shown in Table I .
Cell A cycles between about 20% and 80% charged, which is perfectly satisfactory. However, Cell B sinks lower and lower, and eventually fails after a fairly small number of cycles. If one cell in a battery goes completely flat like this, the battery voltage will fall sharply, because the cell is just a resistance lowering the voltage.
If current is still drawn from the battery, that cell is almost certain to be severely damaged, as the effect of driving current through it when flat is to try and charge it the ‘wrong way'. Because a battery is a series circuit, one damaged cell ruins the whole battery. This effect is probably the major cause of premature battery failure.
Table I Showing the state of charge of two different cells in a battery. Cell A is a good quality cell, with low self-discharge. Cell B has a higher self-discharge, perhaps because of slight manufacturing faults, perhaps because it is warmer. The cells are discharged and charged a number of times
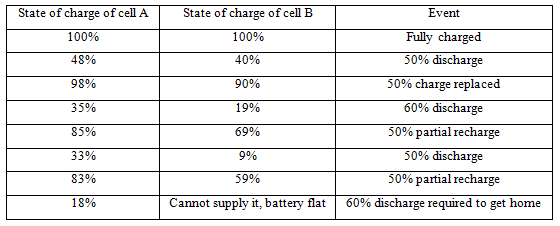
The way to prevent this is to fully charge the battery till each and every cell is fully charged (a process known as charge equalization) at regular intervals. This will inevitably mean that some of the cells will run for perhaps several hours being overcharged. Once the majority of the cells have been charged up, current must continue to be put into the battery so that those cells that are more prone to self-discharge get fully charged up. This is why it is important that a cell can cope with being overcharged.