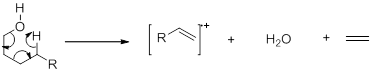
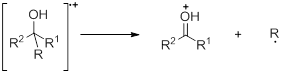
- Analysis of Alcohols : In alcohols the molecular ion may be altogether absent. The predominant fragmentation involves loss of an alkyl group. In case of branched alcohols, the largest alkyl group is usually lost.
-
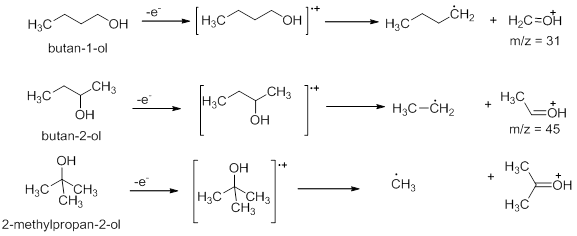
In the spectrum of 1-butanol, the base peak is obtained at m/z =31 due to loss of a propyl group. Butan-2-ol loses an ethyl group to have base peak at m/z = 45. Similarly, 2-methylpropan-2-ol loses a methyl group to give a peak corresponding to m/z = 59.
-
A second common mode of fragmentation is dehydration. Alcohols having four or more carbons may undergo simultaneous loss of ethylene and water.