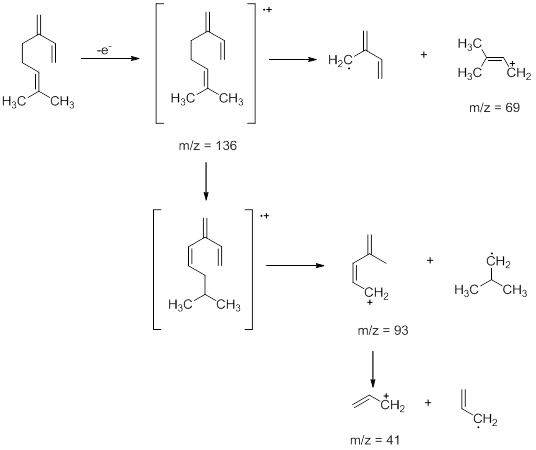
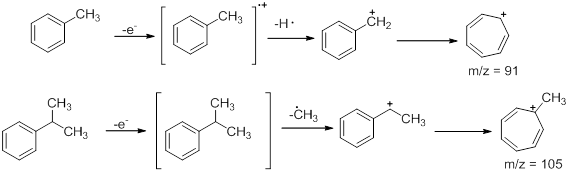
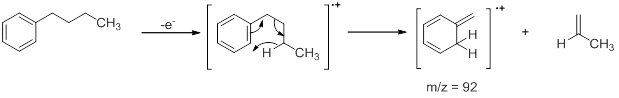
- Aromatic Hydrocarbons : These compounds show a strong molecular ion peak. When an alkyl chain is attached to benzene ring preferential fragmentation occurs at the benzylic position to form a fragment of the formula C7H7+ (m/z =91). Thus, toluene exhibits a peak at 91. The peak at m/z =105 in isopropyl benzenes is also due to a similar fragmentation where one of the benzylic C-H is replaced by methyl group. Similarly, when an alkyl group attached to the benzene ring is a propyl group or larger, Mclafferty rearrangement occurs to give fragments. Using butyl benzene the effect of McLafferty rearrangement can be shown. The peak at m/z = 92 is obtained as a result of McLafferty rearrangement .