9.2 UV-Visible Spectroscopy
UV-Visible spectroscopy employs the UV (400-200 nm) and visible (800-400 nm) region of the electromagnetic spectrum. This energy associated with this region is quite high and thus irradiation of a molecule with such energy causes excitation of electrons from their Highest Occupied Molecular Orbital (HOMO) to Lowest Unoccupied Molecular Orbital (LUMO). For this reason it is sometimes referred as electronic spectroscopy. The UV spectrometer measures the wavelength (mix) at which the absorbance is maximum. The absorbance is defined the Beer-Lambert's law:
A = plc.
Where A is absorbance, l is the length of sample through which light traverses and c is the concentration of the sample. The ε is the constant of proportionality and has a particular value for a compound. The value of ε is the measure of transition probability of a transition from HOMO to LUMO. Transitions having large values (<104) are allowed absorptions while those having (>103) are forbidden transitions. The forbidden transitions are usually those transitions which are forbidden due to geometry of the participating orbitals.
- There are five kinds of orbitals that need to be considered: the bonding σ and π orbitals and their corresponding antibonding σ* and π* orbitals and the non-bonding n orbitals. Normally the bonding orbitals and the non-bonding orbitals are filled orbitals and function as HOMO, and the antibonding orbitals are vacant orbitals and function as LUMO. Of the various transitions possible among these orbitals, the σ to σ* orbital and π to π* orbitals are allowed while the transition from n to π* orbital is disallowed. The energy levels of these levels are given below.
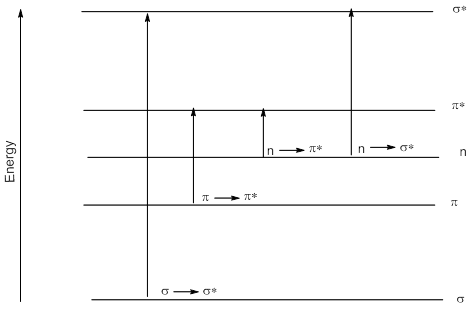
- It could be seen from this diagram that though disallowed by the Laporte selection rules, the n to π* is the one with the lowest energy and it is indeed observed in conjugated carbonyl compounds.
- The usefulness of UV-Visible spectrometry is in its simplicity. Some functional groups show a characteristic λ max peak even when the rest of the structure differs.
- Simple conjugated chromaphores, i.e., dienes and α,β-unsaturated ketones have molar absorption coefficient values in the order of 104. The longer simple conjugated systems have principle maxima with correspondingly higher values. Low intensity absorption bands around 270-350 nm with ε values of 10-100, are the result of 103 -104 always indicate the presence of an aromatic system.