9.3 Mass Spectroscopy
A mass spectrometer determines the molecular weight of a compound. It has three basic components: something to volatilize and ionize the molecule into a beam of charged particles; something to focus the beam so that particles of the same mass: charge ratio is separated from all others; and something to detect the particles.
-
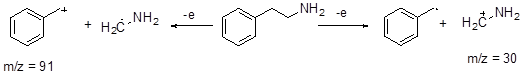
Two methods are usually used to ionize molecule: electron impact and chemical ionization. The former method called electron impact ionization (E.I.) mass spectrometry the molecule is bombarded with highly energetic electrons that knock a weakly bound electron out of the molecule. Losing a single electron leaves behind a radical cation: an unpaired electron and a positive charge. The electron that is lost will be one of relatively high energy, and this will typically be one not involved in bonding, for example, an electron from a lone pair. Thus ammonia gives NH3+• and a ketone gives R2C=O+• . The problem with this technique is that it is too “hard” i.e.; there is a lot of fragmentation which at times makes it difficult to assign the molecular ion peak. In some cases it may be totally absent. Also since the radical cations are extremely unstable they may decompose before reaching the detector. In the later method, chemical ionization, (C.I.) mass spectrometry the electron beam is used to ionize a simple molecule such as methane which in turn ionizes our molecule by collision and transfer of a proton. Under electron bombardment, methane loses a bonding electron to give CH4+• which reacts with an unionized methane molecule to give CH3• and CH5+ . This unstable compound (CH5+) is a powerful acid, and can protonate just about any other molecule. When it protonates our sample, a proton has been added rather than an electron removed, so the resulting particles are simple cations, not radical cations, and are generally more stable than the radical cations produced by direct electron impact. Having more functional groups helps molecular ions to decompose. The aromatic amine 2-phenylethylamine is a brain active amine found in some foods such as chocolate, red wine, and cheese and possibly implicated in migraine. It gives a poor molecular ion by E.I., a base peak with a mass as low as 30 and the only peak at higher mass is a 15% peak at 91. The C.I. mass spectrum on the other hand has a good molecular ion: it is [M + H]+ . Normally a fragmentation gives one cation and another radical, only the cation being detected. It is relatively unusual for one bond to be able to fragment in either direction, but here it does, which means that both fragments are seen in the spectrum.
-
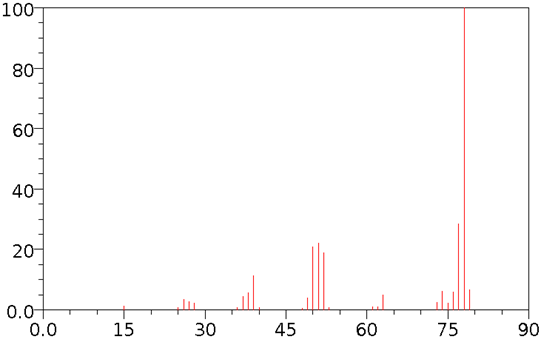
The strongest peak is assigned an ‘abundance' of 100% and called the base peak . In the mass spectrum given below 78 is the base peak. The abundance of the other peaks is shown relative to the base peak. This spectrum is a plot of the abundance of the ions versus mass/ charge (m/z).