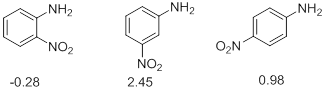
- Electron withdrawing and electron donating mesomeric effects also have influence on the strengths of acids and bases. In the series of o- , m-, p- nitroanilines it is observed that the ortho and para isomers have extremely high acidity as compared to the meta isomer. In fact the o -nitroaniline is a stronger acid than p -nitroaniline. This is explained in terms of electron withdrawing mesomeric effect which is operational only in the ortho and para isomers but not in the meta isomer. The extra base weakening effect, when the substituent is in ortho position in part due to the short distance over which its inductive effect is operating and also to the direct interaction between the nitro and amino group by hydrogen bonding (Scheme 8).
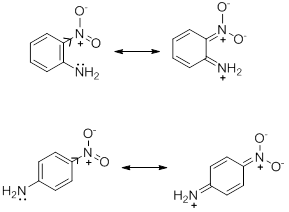
Scheme 8
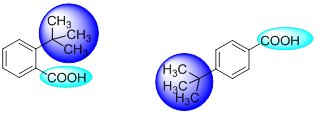
- Steric considerations may also affect the strength of an acid or base. Such an instance is observed in case of o -t-butyl benzoic acid which is ten times stronger than its para isomer. This is totally unexpected since the electron donating inductive effect should be stronger in the ortho isomer compared to the para isomer. However, in this case, the carboxyl group is forced out of the plane of the ring by the bulky t-butyl group in ortho position.
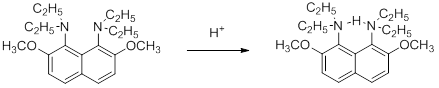
-
Steric effects can also be caused by other types of strain. 1,8-Bis(diethylamino)-2,7-dimethoxynaphthalene is an extremely strong base for a tertiary amine, but proton transfers to and from the nitrogen are exceptionally slow; slow enough to be followed by a UV spectrophotometer. This is because the compound is severely strained and the two nitrogen lone pairs are forced to be near each other. Protonation relieves the strain: one lone pair is now connected to hydrogen, which forms a hydrogen bond to the other lone pair.