- Similarly, substitutions by electron withdrawing groups tend to increase the acidity of an acid. Thus, in the following series of halo acids it is observed that the pKa decreases with increasing number of halogen atoms. Furthermore, it is also observed that among the monosubstituted halo acids the value of pKa decreases with increase in electronegativity of the halogen. This indicates that the more is the electron withdrawing power of a group, the lower is the pKa (Scheme 5).
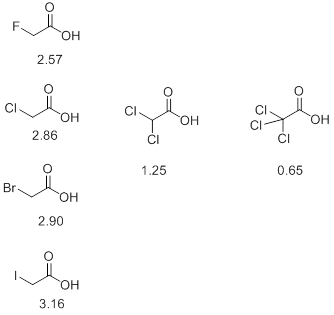
Scheme 5
- In case of aliphatic bases, it is thus expected that the increase in the number of alkyl groups will increase the basicity. Thus, basicity is supposed to increase on going from primary to secondary to tertiary amines. In fact, it is observed that in a solution of water, the basicity does increase on going from the primary amine to secondary amine but drops slightly on going to the tertiary amine. This drop is explained by considering the factor of stabilization of the conjugate acid by solvation. As the number of alkyl groups increase, there is a decrease in the number of hydrogen atoms capable of forming hydrogen bonds with water. This in turn has an adverse effect on the stabilization by solvation. If however, the basicities are measured in solvent where stabilization by solvation is insignificant, then the basicity does increase on going from secondary amine to tertiary amine (Scheme 6).
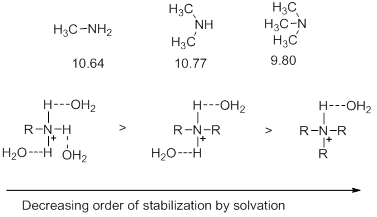
Scheme 6
- There is also an effect of stabilization by delocalization in case of strength of bases (like the case of methanoic acid vs ethanoic acid). Thus, it is found that guanidine is much stronger base than amidine which in turn is stronger than urea. The high basic strength is explained by the formation of 3 identical canonical structures of similar energy for guanidium ion. This results in lowering in energy of the molecule and gives stability to the species. A similar case occurs for amidinium ion where there are two such structures of identical energy. In case of urea, there are no such structures of identical energy and thus it is the least basic of the three (Scheme 7).
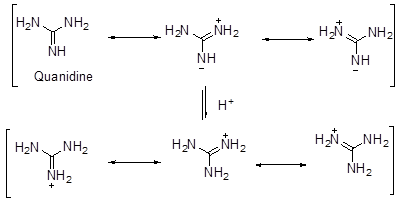
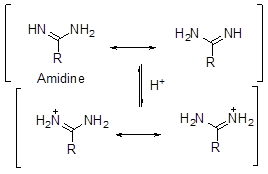
Scheme 7