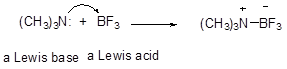2.1 Concepts
A compound is classified as an acid or a base based on certain properties. At present there are several theories which define the concepts of acidity and basicity. Some important concepts are detailed below.
Arrhenius concept of acids and bases : The Arrhenius definition of acids says that they are compounds that give off H+ ions in water and that bases are compounds that give off OH- ions in water. Thus, according to this theory only protic acids are allowed and only hydroxide bases are allowed to be classified as an acid or a base.
![]()
Br o nsted-Lowry definition : The Bronsted-Lowry definition of acids is that acids are compounds that give off H+ ions when they react with another compound. Likewise, this definition says that bases are compounds that accept H+ ions from other compounds. Furthermore, it also brings a new concept of conjugate acids and conjugate bases. Since an acid after donating its proton is technically a base according to this definition and is referred to as a conjugate acid. So every acid has its conjugate base and vice versa. Also, the stronger is an acid, the weaker is its conjugate base and vice versa.
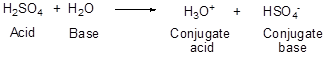
Lewis concept of acids and bases : Acids are electron pair acceptors while bases are electron pair donors. Thus, electron deficient species like BF3 are Lewis acids while electron rich species such as tertiary amines are Lewis bases. Lewis acids may combine with Lewis bases to generate a salt.