2.4 The Effect of Structure on pKa
There are several factors which decide the relative strength of an acid (H-A). The strength of the H-A bond, the electronegativity of A- and the factors stabilizing A- with respect to HA are some of these factors. The first one is usually not much relevant but may be the cause of difference in acid strengths of CH3 OH (16) vs CH4 (43).
-
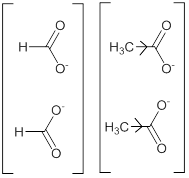
The stabilization of the anion with respect to the undissociated molecule is however of quite importance. As an example, in case of formic acid, the anion generated is greatly stabilized due to effective delocalization of the charge into the two canonical structures of identical energy. Such delocalization is not possible in the formic acid itself without leading to separation of charges (Scheme 3).
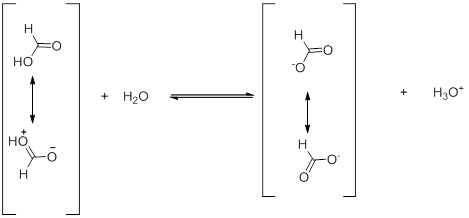
Scheme 3
-
Similarly, the solvent plays an important role. The solvation of the species generated in the solvent could lead to an increase or decrease in acidity. As an acid or base is dissociated, more charges develop which may be better stabilized in solvent having a high dielectric constant and a greater degree of hydrogen bonding ability. Thus, HCl when dissolves in water is very strong acid as the ions it generates are suitably solvated by the water molecules but if the solvent is changed to toluene, there is hardly any dissociation (Scheme 4).
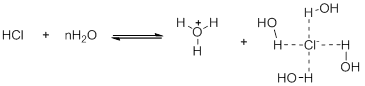
Scheme 4
• It is also expected that electron withdrawing and electron donating groups will have a profound effect on the acid strength of a substance. Thus, electron donating group substituted carboxylic acids are less acidic than those without them. In comparison of the acid strengths of methanoic and ethanoic acids, it is observed that the former has pKa of 3.77 while for the later has only 4.84. The difference has been explained by the electron donating inductive effects of the methyl group of ethanoic acid. However, it is to be noted that the inductive effect falls off quickly with the increase in the chain length and other factors become dominant on going down the series.