2.2 pH and pKa
The strength of an acid depends on its degree of dissociation in water and can be determined from the equilibrium expression (Scheme 1).
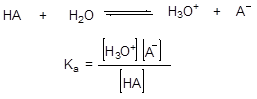
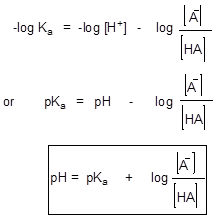
Scheme 1
This equation is known as Hendersson-Hasselbach equation and relates a relationship between the concentration of hydrogen ions in solutions and acid strength. Thus, pH is a measurement of the H+ concentration in a liquid. If there is a high H+ concentration, the pH indicates that it is a very acidic solution. If the solution is neutral, there is only a small H+ concentration, and the pH reflects that. If the solution is basic, there is almost no H+ concentration, and you can tell that by the pH number.
As with acids, a similar scale may be derived for bases based on the equilibrium of the protonated form of the base and base itself (Scheme 2).
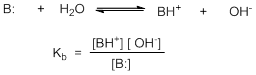
Scheme 2
However, such an expression creates two separate scales for the determination of strengths of acids and bases. Instead the strength of the conjugate acid is determined then a common scale in terms of pKa may be used for both acids and bases. Thus, the lower the value of pKa, stronger is the acid. Similarly, higher is the value of pKa of base, more basic it is.
2.3 Buffer Solution
A solution of a weak acid and its conjugate base is referred as a buffer solution. A buffer solution has the ability to resist to a small change of pH, because the weak acid can donate a proton to any HO- added to the solution, and its conjugate base can accept any H+, which is added to the solution.
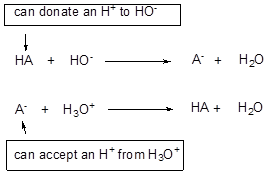
A common example of buffer is a solution of sodium acetate in acetic acid. Human blood is also a buffered solution and maintains a pH close to neutrality.